A fraction of activated B cells become quiescent memory B cells. In subsequent encounters with the same antigen and again with memory Th cells against that same antigen, the memory B cells respond with a much shorter lag period and produce more antibodies for a longer period of time than in the primary response of the naïve Th and B cells. In addition, this secondary response produces antibodies with higher affinity, primarily of the IgG isotype.
Links between the Innate and Adaptive Defense Systems
Treating the innate and adaptive defense systems as separate subjects in separate chapters makes pedagogical sense because it is generally easier to understand complex systems by first separating them into their constituent components. However, it is important to note that for the human body to have the innate defenses acting independently of the adaptive defenses would be counterproductive, because the two types of defense need to act synergistically. Some examples of links are cytokines and chemokines, which are produced by and act on both types of immune responses; PMNs that serve as first responders and contribute through cytokine production to the Th1/Th2 decision-making process; DCs that opsonize pathogens and present the processed antigens to the adaptive immune cells; and antibodies that are made as an adaptive response but, when bound to bacterial cells, activate one arm of the complement cascade (i.e., the classical pathway).
We now add yet another link: the interaction between natural killer cells and B cells. It may be simply a coincidence that natural killer (NK) cells have a number of similarities, morphologically and cytokinologically, to T cells. Because T cells interact with B cells on other planes of existence, it is somewhat satisfying to learn that NK cells interact with B cells, too. Briefly, NK cells can stimulate B cells to proliferate (as T helper cells do) but by some factor that is not one of the known cytokines. B cells in turn induce NK cells to secrete more IFN-γ, which, as we have already seen, is an important factor in macrophage activation.
T-Cell-Independent Antibody Responses
In addition to the CD1-dependent T cell pathway discussed earlier ( Figure 4-6C), nonpeptide antigens such as polysaccharides (e.g., bacterial capsule components), nucleic acids (e.g., bacterial DNA or RNA), and to a lesser extent lipids and glycolipids (e.g., bacterial membrane components) can also stimulate T-cell-independent antibody responses. T-cell-independent antigens provoke an antibody response by directly interacting with B cells ( Figure 4-11), without the involvement of T cells. Polysaccharides are a well-studied class of T-cell-independent antigens. Those B cells displaying mIg with affinity for the sugar repeat units will bind to the polysaccharide chain. Because these epitopes are repeated many times, numerous mIgs and BCRs are linked together, which causes them to cluster and activate cellular signaling pathways. This acts as one signal to stimulate the B cells to proliferate and differentiate into mature antibody-producing plasma cells. A second signal, such as binding of cytokines produced at the infection site, is also required to initiate the T-cell-independent response.
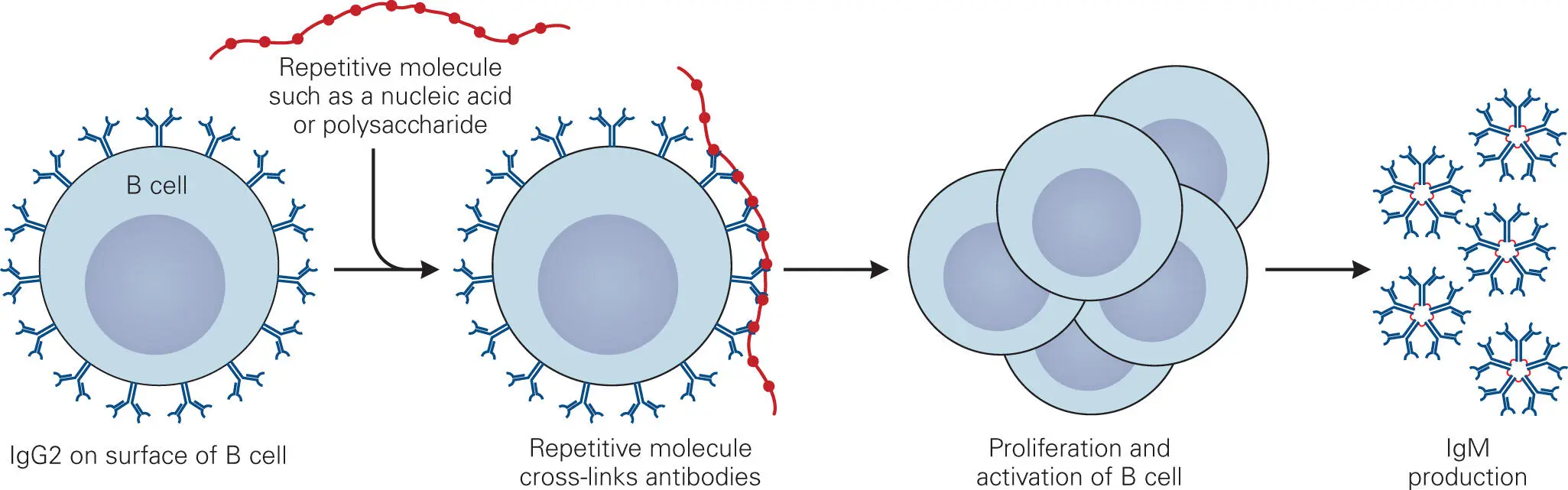
Figure 4-11. T-cell-independent production of antibodies. Macromolecules with highly repetitive structures, such as carbohydrates (polysaccharides, capsules) and nucleic acids, are able to directly stimulate naïve B cells to proliferate, mature, and produce antibodies through antigen-mediated clustering of IgG2 and IgM antibodies on the surface of the B cells.
The T-cell-independent response is particularly important for protection against bacterial pathogens that can avoid phagocytosis by covering themselves with an exopolysaccharide layer (capsule), which is not effectively opsonized through coating with C3b. Such bacterial pathogens are only ingested and killed by phagocytes if antibodies that bind to capsular antigens are elicited and act as opsonins.
Although the T-cell-independent response provides protection against capsule-producing bacteria, it has some important drawbacks. First, the antibody response elicited by T-cell-independent antigens is not as strong as the T-cell-dependent response. It is also not long-lasting, because no memory B cells are developed. Second, the main antibodies elicited by T-cell-independent antigens are IgM and IgG2. IgG2 does not opsonize and IgM does so less effectively than IgG1 and IgG3 ( Table 4-1). Third, infants do not mount a T-cell-independent response. Polysaccharides and lipids can elicit an immune response in children and adults, but not in infants under the age of two years. Thus, the ability to respond to T-cell-independent antigens, such as capsule exopolysaccharide, is acquired after birth. This is an important consideration because it means that vaccines consisting of T-cell-independent antigens are not effective until an infant has become old enough to respond to these antigens. Unfortunately, infants are one of the highest risk groups for contracting serious infections due to capsule-producing bacteria (e.g., pneumonia and meningitis).
A strategy for improving the immune response to T-cell-independent antigens and extending this response to infants is to covalently link epitopes of the polysaccharide capsule to a protein. This type of vaccine is called a conjugate vaccine (more about this in chapter 17). APCs process conjugate vaccines as if they were proteins and elicit a Th2-cell-dependent response that culminates in production of antibodies that also recognize the polysaccharide antigens. This immune response is long-lived because it involves T cell activation and memory cell generation and produces opsonizing IgG1 antibodies, IgG3, and IgG4.
Mucosal Immunity: IgA/sIgA Antibodies
An important immune defense against infectious diseases, but one that is much less well understood than the humoral or cell-mediated responses, is the immune system that produces sIgA. The first step in many microbial infections is colonization and invasion of a mucosal surface. sIgA can prevent such infections by blocking colonization. Thus, while the cell-mediated or humoral antibody responses may cause collateral damage to tissues in the area where infection is occurring, the sIgA-mediated defense is usually innocuous to the host because it occurs in the mucus layer.
As we mentioned in chapter 2, skin and mucosal surfaces all have attendant mucosa-associated lymphoid tissues (MALT) located just below the epithelial layer in the lamina propria. The mucosal surface of the small intestine is underlain with the GALT, while the lungs have BALT, the upper respiratory tract has NALT, and the vaginal tract has vaginal-associated lymphoid tissue (VALT). Skin also has a similar system (called SALT).
The intestinal cells that comprise the GALT are visible as a collection of follicles, called Peyer’s patches, which are most highly concentrated in the ileum and rectum of the intestine. Similar mucosal lymphoid tissues are found in the respiratory and vaginal tracts, although they are not as pronounced. The cells that form the Peyer’s patches are illustrated in Figure 4-12. M (microfold) cells take up antigens from the lumen of the intestinal tract and pass them to closely associated GALT macrophages, which act as the APCs of the GALT. M cells have never been successfully cultivated in vitro, so little is known about the activities of M cells. The mechanism by which GALT macrophages process antigens and elicit production of cytotoxic T cells or antibodies is the same as that described in earlier sections, except that the macrophages, B cells, and T cells of the GALT reside specifically in the lamina propria of mucosal surfaces. Two additional types of T cells, Th17 and Tregs, appear to be important for IgA production in the intestine, both of which appear to help modulate the B cells.
Читать дальше













