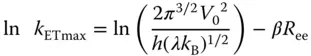( 3.2) 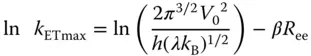
where V 0refers to the maximal electronic coupling element, while β is the decay coefficient factor (damping factor), which depends primarily on the nature of the bridging molecule. From the linear plot of ln k ETmaxvs. R eethe β value is obtained as 0.60 Å −1[39]. This β value is located within the boundaries of nonadiabatic electron transfer reactions for saturated hydrocarbon bridges (0.8–1.0 Å −1) and unsaturated phenylene bridges (0.4 Å −1) [43].
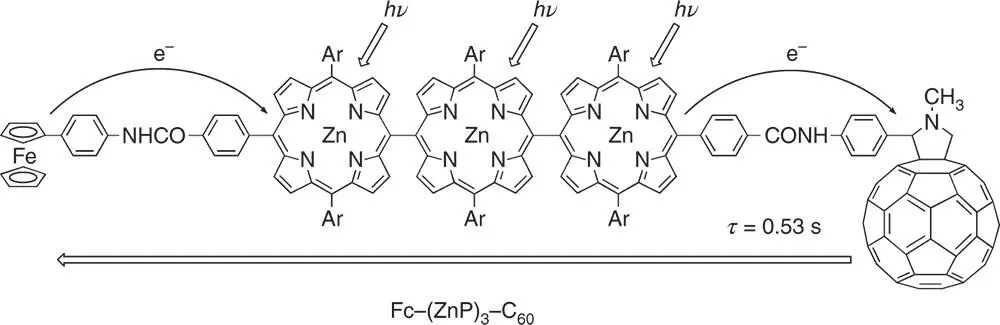
Scheme 3.1Multistep photoinduced electron transfer in a ferrocene‐ meso , meso ‐linked porphyrin trimer–fullerene pentad (Fc–(ZnP) 3–C 60); Ar = 3,5‐Bu t 2C 6H 3.
Source: Imahori et al. 2004 [44]. Reproduced with permission of John Wiley & Sons.
The best molecule mimicking multistep electron‐transfer processes in the photosynthetic reaction center so far reported is a ferrocene‐ meso , meso ‐linked porphyrin trimer–fullerene pentad (Fc–(ZnP) 3–C 60in Scheme 3.1), where the C 60and the ferrocene (Fc) are tethered at both the ends of (ZnP) 3( R ee= 46.9 Å) [44]. The lifetime of the final CS state (0.53 seconds at 163 K) has been attained without lowering the CS efficiency (Φ = 0.83) [44].
4 Electron Donor–Acceptor Dyads
Multistep electron‐transfer processes have been utilized to attain a long distance charge separation (CS), mimicking the natural photosynthetic reaction center (vide supra). However, a significant amount of energy is lost during the multistep electron‐transfer processes to reach the final CS state [11–15]. In photosynthesis a two–step photoexcitation, the so‐called “Z‐scheme,” is thereby required to recover the energy loss via the multistep electron‐transfer processes and to gain strong oxidizing power to oxidize water as well as high reducing power to reduce NAD +coenzyme [4]. The design and synthesis of molecular machinery mimicking such an elaborated “Z‐scheme” in nature seems far beyond our capability, and even if it could be done, the synthetic cost would certainly preclude any type of practical application. Thus, it is highly desired to design simple molecular electron donor–acceptor dyads that are capable of fast CS but can retain slow charge recombination (CR). Theoretically, it is possible to obtain such an electron donor–acceptor dyad, because the CS lifetime increases with increasing driving force of electron transfer in the Marcus inverted region (vide supra). However, the driving force of electron transfer should be lower than the triplet excited state of one of the components of donor–acceptor dyads. Otherwise, the CS state would decay rapidly to the triplet excited state in the Marcus normal region rather than to the ground state in the Marcus inverted region [35].
A number of simple donor–acceptor dyads have been designed and synthesized to attain long‐lived CS state, where the donor and acceptor molecules are linked with a short spacer to minimize the solvent reorganization energy [45–50]. Efficient photoinduced electron transfer occurs in a zinc imidazoporphyrin–C 60dyad (ZnImP–C 60) with a short linkage to form the CS state (ZnImP ·+–C 60 ·−) with the rate constant of 1.4 × 10 10s −1( Scheme 4.1) [45]. The CS state (1.34 eV) is lower in energy than both the triplet excited states of C 60(1.50 eV) and ZnImP (1.36 eV) [45]. The CS state, produced upon photoexcitation of ZnImP–C 60, is detected by the transient absorption spectrum, which has absorption bands at 700 and 1040 nm due to ZnImP ·+and C 60 ·−, respectively [45]. The CS state decays by back electron transfer to the ground state, obeying first‐order kinetics with a rate constant of 3.9 × 10 3s −1(the lifetime is 260 μs) at 298 K [45]. At 278 K the lifetime of the CS state was determined as 310 μs, which is much longer than those of conventional donor–acceptor dyads with longer spacers [7–9].
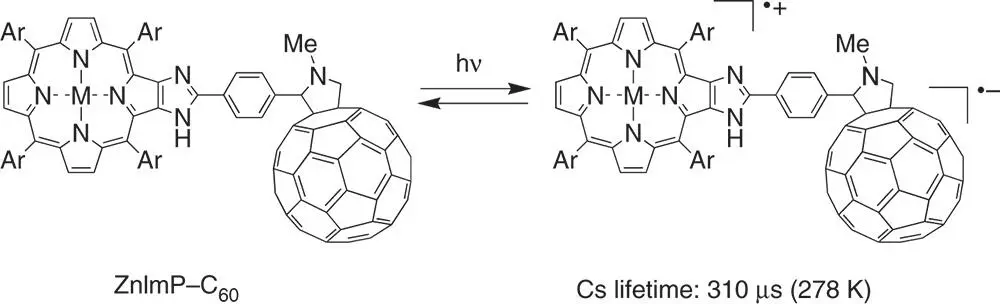
Scheme 4.1Formation of a long‐lived CS state of a zinc imidazoporphyrin–C 60dyad (ZnImP–C 60) with a short linkage (Ar = 3,5‐Bu t 2C 6H 3).
Source: Kashiwagi et al. 2003 [45]. Reproduced with permission of American Chemical Society.
An electron donor–acceptor dyad linked with a short spacer containing Au(III) and Zn(II) porphyrins (ZnPQ–AuPQ +in Scheme 4.2) also affords a long‐lived electron‐transfer state with a lifetime of 10 μs in nonpolar solvents such as cyclohexane [46]. The introduction of quinoxaline to the gold porphyrin results in a lowering of the electron‐transfer state energy. In contrast to the case of neutral donor–acceptor dyads, the energy of the electron‐transfer state (ZnPQ ·+–AuPQ) becomes smaller in a less polar solvent, which is lower than the energies of the triplet excited states of ZnPQ (1.32 eV) and AuPQ +(1.64 eV) [46]. Photoinduced electron transfer occurs from the singlet excited state of the ZnPQ ( 1ZnPQ *) to the metal center of the AuPQ +moiety to produce ZnPQ ·+–Au IIPQ. The observed long lifetime of ZnPQ ·+–Au IIPQ results from a small reorganization energy for the metal‐centered electron transfer of AuPQ +in nonpolar solvents due to the small change in solvation upon electron transfer as compared with that in polar solvents [46]. In a polar solvent such as benzonitrile (PhCN), no CS state was observed, but instead only the triplet–triplet absorption due to 3ZnPQ *–AuPQ +was observed [46]. The absence of an observable CS state in PhCN is ascribed to the much slower photoinduced electron transfer due to the large reorganization energy as compared with that in nonpolar solvents allowing an efficient intersystem crossing process in the ZnPQ–AuPQ +dyad to produce the triplet excited state 3ZnPQ *–AuPQ +[46].
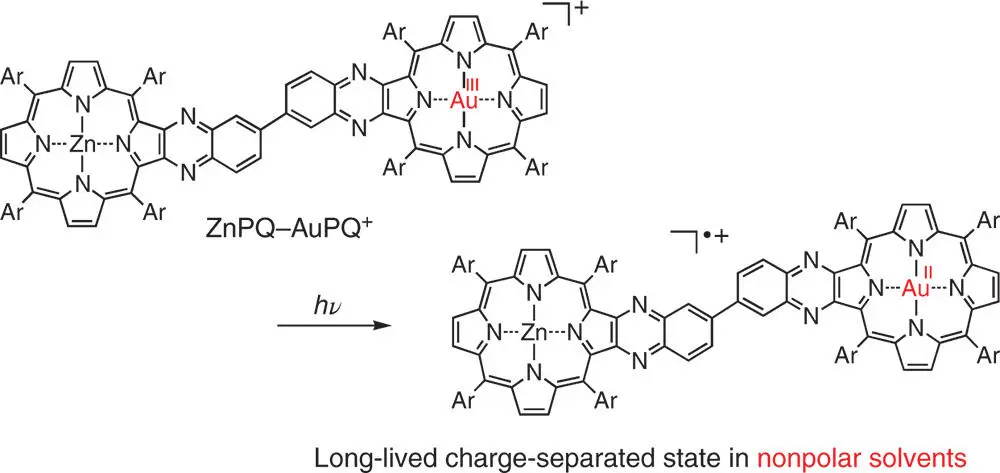
Scheme 4.2Formation of a long‐lived CS state of ZnPQ–AuPQ +in nonpolar solvents (Ar = 3,5‐Bu t 2C 6H 3).
Source: Fukuzumi et al. 2003 [46]. Reproduced with permission of American Chemical Society.
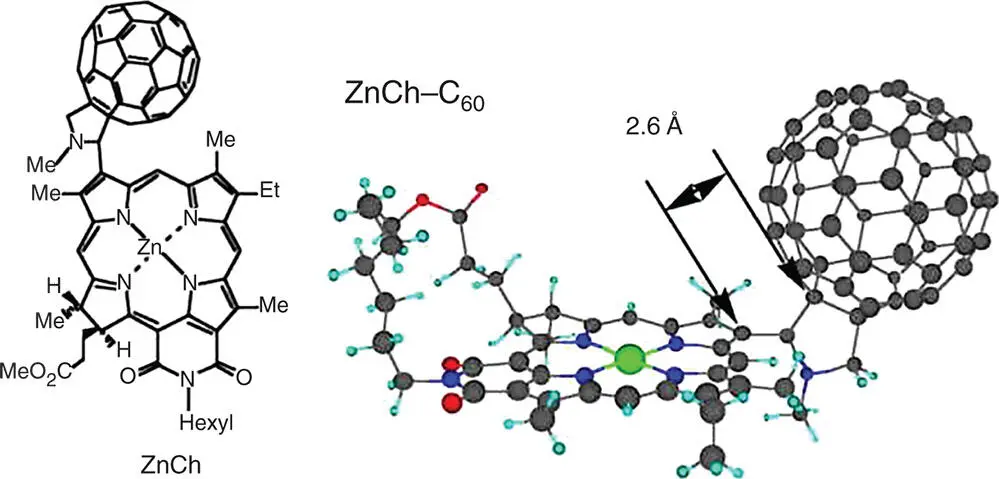
Figure 4.1Structure of a closely linked ZnCh–C 60dyad.
Source: Ohkubo et al. 2004 [47]. Reproduced with permission of John Wiley & Sons.
A closely linked zinc chlorin–fullerene dyad (ZnCh–C 60in Figure 4.1) affords a longer CS lifetime as compared with other zinc chlorin–fullerene dyads with longer spacers [47–51]. A deoxygenated PhCN solution containing ZnCh–C 60gives rise upon a 388 nm laser pulse to a transient absorption maximum at 460 nm due to the singlet excited state of ZnCh [47]. The decay rate constant was determined as 1.0 × 10 11s –1, which agrees with the value determined from the fluorescence lifetime measurements [47]. The decay of absorbance at 460 nm due to 1ZnCh *is accompanied by an increase in absorbance at 590 nm due to ZnCh ·+[47]. This indicates that electron transfer from 1ZnCh *to C 60occurs rapidly to form the CS state, ZnCh ·+–C 60 ·–. The CS state decays via back electron transfer to the ground state rather than to the triplet excited state, because the CS state is lower in energy (1.26 eV) than the triplet excited states of both C 60(1.50 eV) and ZnCh (1.36–1.45 eV) [47]. The lifetime of the CS state is determined as 230 μs at 298 K. The large temperature dependence of the CS lifetime is observed and the lifetime of the CS state at 123 K becomes as long as 120 seconds [47].
Читать дальше