Last, and certainly not least, we come to the nucleic acids.
First, consider the most well-known of these molecules, DNA or deoxyribonucleic acid. This molecule is a complex assembly, but when stripped down, it is beautifully simple. It takes up the three-dimensional structure of a right-handed double helix, but let's untwist this and flatten it down on the table and take a close look at it. First, we have the two backbones that run down the length of the molecule. They are made up of a pentose (deoxyribose) sugar attached to a phosphate group that repeats along the length of the molecule as a sugar–phosphate backbone. You can think of this as scaffolding. It has an important role to play in holding the DNA molecule together and mediating the interactions between DNA and other molecules. The backbone imparts a repeating negative charge to the molecule that helps keep it within the cell since it is repelled by negative charges in the cell membrane.
The information storage capacity of DNA is made possible by the molecules in its center. The pentose sugar of the backbone is attached to a nucleobase through a glycosidic link. There are four nucleobases(sometimes just called bases) in DNA: thymine (T), adenine (A), guanine (G), and cytosine (C) (Figure 4.13a). Let us call these bases the “letters” of the DNA. These letters are the genetic code, and we can attach them to the sugar–phosphate backbone to generate long sequences of them. It is this sequence of letters that codes the instructions for all biological functions, as shall become apparent later. Thymine and cytosine are also referred to as pyrimidines, a class of molecules possessing a single aromatic ring (Figure 4.13a). Adenine and guanine are purines, a class of molecule with both a pyrimidine ring and an imidazole ring (or heterocyclic ring because it is a ring that contains different elements, C and N in this case; Figure 4.13a).
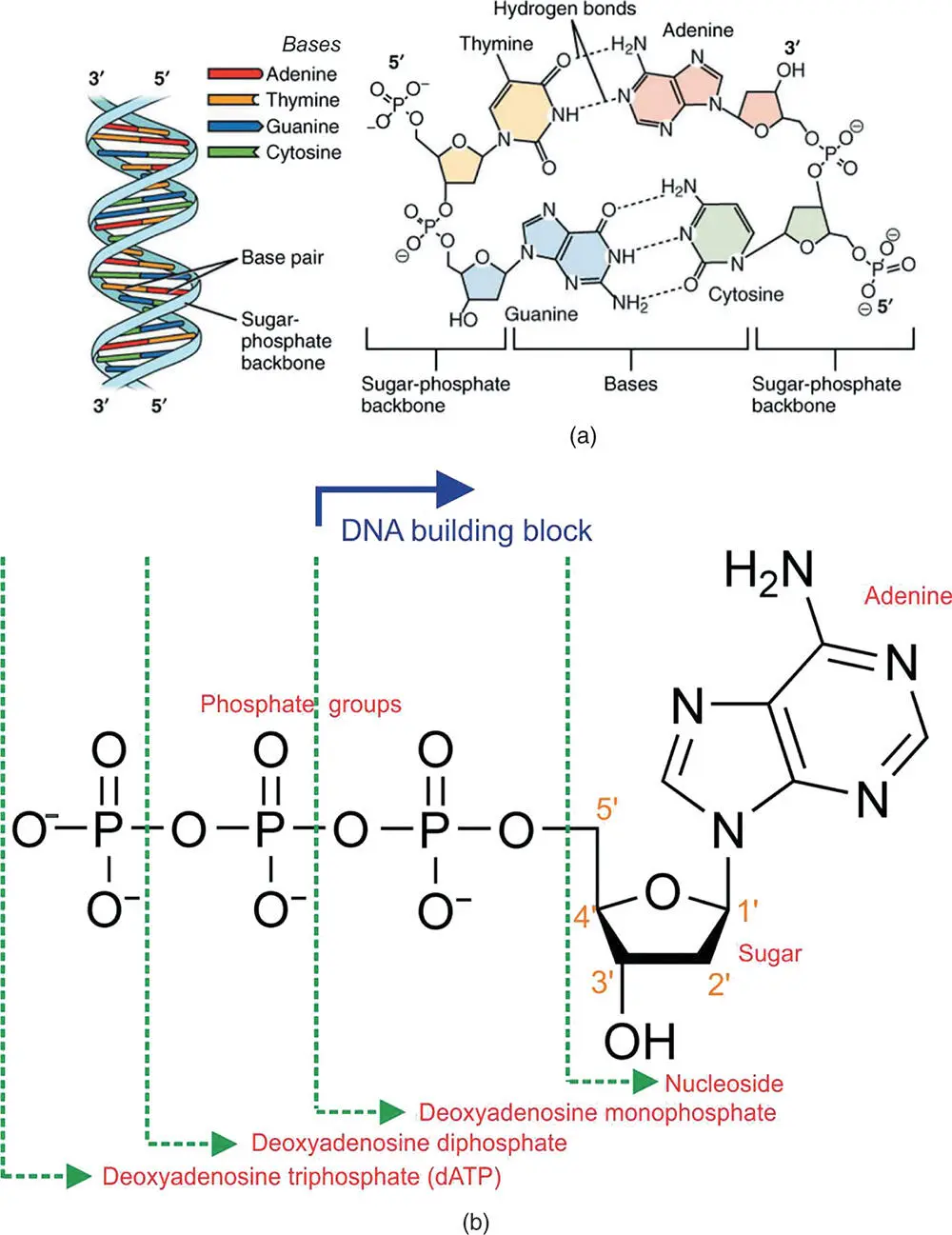
Figure 4.13 The structure of DNA and its building blocks. (a) The structure of DNA showing the double helical structure and base pairing between the bases along the center of the molecule.
Source: OpenStax College.
This is the most common “B” form of the molecule (b) the structure of deoxyadenosine triphosphate, the starting molecule from which an adenosine is inserted into a growing DNA chain. Also shown are the names given to component parts of this molecule. In the DNA chain, one phosphate group and the sugar contribute to the growing backbone, while the base is incorporated into the center of the helix to form part of the DNA code. The numbering convention on the deoxyribose sugar is also shown. This accounts for the directionality in DNA, expressed as the 3′ and 5′ ends.
There is a very important subtlety to be understood here. In DNA, the pentose sugar lacks the hydroxyl group (–OH) on the 2′ position (Figure 4.13). This is why the sugar is called “deoxyribose” and the entire molecule is called deoxy ribonucleic acid. In its sister molecule, RNA(discussed in the next section), this –OH group is present. The consequences of this are that the building blocks of DNA are referred to with the prefix “deoxy-,” for example deoxyadenosine triphosphate (dATP) (Figure 4.13). In advance, you might like to compare this to the structure of ATP shown in Chapter 6(Figure 6.2) that has a hydroxyl group in the 2′ position. In the cell, ATP is processed into dATP prior to DNA synthesis.
In Chapter 5, we discuss DNA replication in more detail, but in the context of this chapter, it is useful to explain how the chains of DNA are assembled from the molecular point of view. When DNA synthesis occurs, the last two phosphate groups on the dATP (Figure 4.13) are cleaved to yield a molecule with the base, the sugar, and one phosphate group (a DNA building block), with the release of energy. This molecular unit joins the extending DNA chain, with the release of the two phosphate groups (called “pyrophosphate”) into solution. Each DNA base has a corresponding triphosphate molecule (dATP, dCTP, dGTP, and dTTP), so that the four of them are used to synthesize the growing DNA chain with its four-letter code. Figure 4.13 only shows the structure of the building blocks for adenine.
There is some other nomenclature worth mentioning. The base and its attached sugar are called a nucleoside(Figure 4.13b). If we then add at least one phosphate group, we use the term nucleotide. You will often see the building blocks of DNA referred to as the nucleotides.
The DNA molecule has two backbones. How is this explained? Through the center of the molecule, each base is hydrogen-bonded to another base in a very specific bonding pattern. The A binds to a T and vice versa, and the G binds to a C and vice versa. We end up with two DNA strands bound through their center with hydrogen bonds. You can probably see why this is important. It certainly occurred to Francis Crick (1916–2004) and James Watson (1928–) in 1953 when they discovered and first published this structure. If we unzip the molecule along the hydrogen bonds, each strand can be used to re-assemble the complementary strand because, as we already know, each base can only bind very specifically to another base. In this way, DNA can be replicated. The two separate DNA strands can be used to recreate two individual new DNA double helices.
It is also important to know that a DNA strand has a direction. It runs from the 3′ (pronounced “three prime”) to the 5′, where these numbers are defined by the numbering convention on the deoxyribose sugar on the DNA backbone. The 3′ carbon has the hydroxyl group attached to it, and the 5′ carbon has the phosphate group attached to it. You can see this numbering convention in Figure 4.13b and the labeling of the strands according to this convention in Figure 4.13a. The two strands of DNA that make the double helix run in opposite directions are said to be “anti-parallel.”
DNA can be packed in slightly different forms. The most common form in the cell, and the one that most of us are familiar with, is the “B” form of the right-handed helix. The B form has a well-defined distance of 0.34 nanometers (nm) between the base pairs, 3.4 nm per turn of the helix, and there are about 10 base pairs per turn of the helix. This helix is 1.9 nm in diameter. Another form of DNA is the intriguing “Z” form of the molecule. This is a left-handed helix, which tends to form when there are alternating stretches of purines and pyrimidines (e.g. CGCGC). Only a small amount of DNA in a cell exists in the Z form. It is not known if this is a serendipitous result of the bases causing this structural alteration or whether the structure is involved in the regulation of any cellular functions.
Another form of nucleic acid is the “A” form, which is a tighter right-handed helix found when DNA binds to strands of RNA, or when two RNA molecules bind to form a helix. We now examine the RNA molecule.
DNA is not the only type of nucleic acid. Another important type is RNA. It shares the same fundamental structure as DNA, but has some crucial differences (Figure 4.14). First, the ribose sugar has a hydroxyl (–OH) group on the 2′ position on the sugar, unlike the sugar in DNA (deoxyribose), hence its name ribonucleic acid, as opposed to deoxy ribonucleic acid (DNA). Second, in RNA the thymine base is replaced with a uracil (U) base. Uracil is a demethylated form of thymine (it lacks the methyl, or CH 3group, on the ring).
Читать дальше













