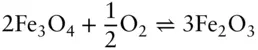Now we need to check our simplifying assumption that we could neglect OH –. The equilibrium between OH –and H +is given by:
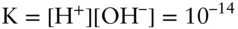
From this we compute [OH –] as 10 −10in the first case and 10 −11in the second. Including these would not change the anion sum significantly, so our assumption was justified.
| Charge balance for rainwater |
|
Rain 1 |
Rain 2 |
| Σ cations |
38 |
204 |
| Σ anions |
63 |
1057 |
| Δ |
25 |
853 |
| pH |
4.60 |
3.07 |
3.10.4 Equilibrium constant expressions
For each chemical reaction in our system, we can write one version of eqn. 3.85. This allows us to relate the equilibrium activities of the species undergoing reaction in our system to one another.
Solution of aqueous equilibria problems often hinge on the degree to which we can simplify the problem by minimizing the number of equilibrium constant expressions we must solve. For example, H 2SO 4will be completely dissociated in all but the most acidic natural waters, so we need not deal with reactions between H +, SO 4 2–, HSO 4 –, and H 2SO 4, and need not consider the latter two in our list of species. Similarly, though many natural waters contain Na +and Cl –, NaCl will precipitate only from concentrated brines, so we generally need not consider reactions between NaCl, Na +, and Cl –.
Carbonate is a somewhat different matter. Over the range of compositions of natural waters, H 2CO 3, 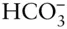 , and
, and 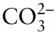 may all be present. In most cases, however, one of these forms will dominate and the concentrations of the remaining ones will be an order of magnitude or more lower than that of the dominant one. In some cases, two of the above species may have comparable concentrations and we will have to consider equilibrium between them, but it is rarely necessary to consider equilibrium between all three. Thus, at most we will have to consider equilibrium between H 2CO 3and
may all be present. In most cases, however, one of these forms will dominate and the concentrations of the remaining ones will be an order of magnitude or more lower than that of the dominant one. In some cases, two of the above species may have comparable concentrations and we will have to consider equilibrium between them, but it is rarely necessary to consider equilibrium between all three. Thus, at most we will have to consider equilibrium between H 2CO 3and 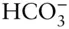 , or
, or 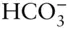 and
and 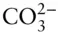 , and we can safely ignore the existence of the remaining species. A successful solution of problems involving carbonate equilibria often requires correctly deciding which reactions to ignore. We will discuss carbonate equilibrium in greater detail in Chapter 6.
, and we can safely ignore the existence of the remaining species. A successful solution of problems involving carbonate equilibria often requires correctly deciding which reactions to ignore. We will discuss carbonate equilibrium in greater detail in Chapter 6.
3.11 OXIDATION AND REDUCTION
An important geochemical variable that we have not yet considered is the oxidation state of a system. Many elements exist in nature in more than one valence state. Iron and carbon are the most important of these because of their abundance. Other elements, including transition metals such as Ti, Mn, Cr, Ce, Eu, and U, and nonmetals such as N, S, and As, are found in more than one valence state in nature. The valence state of an element can significantly affect its geochemical behavior. For example, U is quite soluble in water in its oxidized state, U 6+, but is much less soluble in its reduced state, U 4+. Many uranium deposits have formed when an oxidized, U-bearing solution was reduced. Iron is reasonably soluble in reduced form, Fe 2+, but much less soluble in oxidized form, Fe 3+. The same is true of manganese. Thus, iron is leached from rocks by reduced hydrothermal fluids and precipitated when these fluids mix with oxidized seawater. Eu 2+in magmas substitutes readily for Ca in plagioclase, whereas Eu 3+does not. Nitrogen is the element most critical to life after C, H, and O and is abundant in the atmosphere and dissolved in natural waters as N 2. Plants and algae, however, can utilize N only in its reduced or oxidized states (such as ammonium or nitrate). The mobility of pollutants, particularly toxic metals, will depend strongly on whether the environment is reducing or oxidizing. Thus, the oxidation state of a system is an important geochemical variable.
The valence number of an element is defined as the electrical charge an atom would acquire if it formed ions in solution. For strongly electronegative and electropositive elements that form dominantly ionic bonds, valence number corresponds to the actual state of the element in ionic form. However, for elements that predominantly or exclusively form covalent bonds, valence state is a somewhat hypothetical concept. Carbon, for example, is never present in solution as a monatomic ion. Because of this, assignment of valence number can be a bit ambiguous. A few simple conventions guide assignment of valence number:
The valence number of all elements in pure form is 0.
The sum of valence numbers assigned to atoms in molecules or complex species must equal the actual charge on the species.
The valence number of hydrogen is +1, except in metal hydrides, when it is −1.
The valence number of oxygen is −2 except in peroxides, when it is −1.
The valence state in which an element will be present in a system is governed by the availability of electrons. Oxidation–reduction (redox) reactions involve the transfer of electrons and the resultant change in valence. Oxidation is the loss of electrons; reduction is the gain of electrons . ‡An example is the oxidation of magnetite (which consists of 1 Fe 2+and 2 Fe 3+) to hematite:
(3.101) 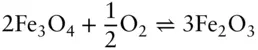
The Fe 2+in magnetite loses an electron in this reaction and is thereby oxidized; conversely, oxygen gains an electron and is thereby reduced.
We can divide the elements into electron donors and electron acceptors ; this division is closely related to electronegativity, as you might expect. Electron acceptors are electronegative; electron donors are electropositive. Metals in 0 valence state are electron donors, nonmetals in 0 valence state are usually electron acceptors. Some elements, such as carbon and sulfur, can be either electron donors or receptors. Oxygen is the most common electron acceptor, hence the term oxidation . It is nevertheless important to remember that oxidation and reduction may take place in the absence of oxygen.
A reduced system is one in which the availability of electrons is high, due to an excess of electron donors over electron acceptors. In such a system, metals will be in a low valence state (e.g., Fe 2+). Conversely, when the availability of electrons is low, due to an abundance of electron acceptors, a system is said to be oxidized. Since it is the most common electron acceptor, the abundance of oxygen usually controls the oxidation state of a system, but this need not be the case.
To predict the equilibrium oxidation state of a system, we need a means of characterizing the availability of electrons, and the valence state of elements as a function of that availability. Low-temperature geochemists and high-temperature geochemists do this in different ways. The former use electrochemical potential while the latter use oxygen fugacity. We will consider both.
Читать дальше

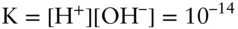
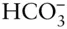 , and
, and 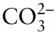 may all be present. In most cases, however, one of these forms will dominate and the concentrations of the remaining ones will be an order of magnitude or more lower than that of the dominant one. In some cases, two of the above species may have comparable concentrations and we will have to consider equilibrium between them, but it is rarely necessary to consider equilibrium between all three. Thus, at most we will have to consider equilibrium between H 2CO 3and
may all be present. In most cases, however, one of these forms will dominate and the concentrations of the remaining ones will be an order of magnitude or more lower than that of the dominant one. In some cases, two of the above species may have comparable concentrations and we will have to consider equilibrium between them, but it is rarely necessary to consider equilibrium between all three. Thus, at most we will have to consider equilibrium between H 2CO 3and 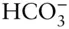 , or
, or 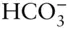 and
and 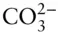 , and we can safely ignore the existence of the remaining species. A successful solution of problems involving carbonate equilibria often requires correctly deciding which reactions to ignore. We will discuss carbonate equilibrium in greater detail in Chapter 6.
, and we can safely ignore the existence of the remaining species. A successful solution of problems involving carbonate equilibria often requires correctly deciding which reactions to ignore. We will discuss carbonate equilibrium in greater detail in Chapter 6.