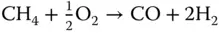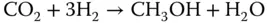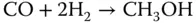Table 1.2Recent shale gas reserves and production in for the six countries with more shale gas reserves.
Source: From EIA 2015 [13].
| Country |
Unproved recoverable reserves by 2013 (Tcf) |
Production in 2018 (Bcf/yr) |
References |
| China |
1115.20 |
353.15 |
[8] |
| Argentina |
801.50 |
365.00 |
[9] |
| Algeria |
706.90 |
No production |
[10] |
| United States |
662.50 (by 2015) |
7079.62 |
[4] |
| Canada |
572.90 |
182.80 |
[11] |
| Mexico |
545.20 |
No production |
[12] |
1.3 Shale Gas Composition
One particular characteristic of shale gas is its varying composition. Shale gas composition depends heavily on the location of the sources, and it may variate even within wells in the same play. The primary component of shale gas is methane, but it also contains considerable quantities of natural gas liquids (NGLs) such as ethane and propane. Apart from these components, shale gas also contains acid gases such as CO 2, H 2S, and inorganic components such as nitrogen [5,14]. The separation of NGLs from methane has induced industries to look for alternatives to transform them into more valuable products, but at the same time the varying composition of shale gas represents a challenge for the treatment plants, which have to be robustly designed to handle such variations in the gas composition.
1.4 Shale Gas Effect on Natural Gas Prices
The high availability of natural gas, generated as a result of the increasing production of shale gas, has caused a noticeable drop of its price in the United States. Moreover, the ability to extract natural gas from deposits that are not associated to crude oil reservoirs has uncoupled natural gas and crude oil prices [1]. These facts have contributed to what has been defined as the new era of cheap natural gas, in which it has been priced consistently under US$5 per million Btu for almost a decade in the United States [15]. In particular, natural gas prices in 2019 have shown a decrease from 3.18 at the beginning of the year to US$2.07 per million Btu in September [16]. Even more, in an extreme situation, producers at the Waha hub in the Permian basin in West Texas had to pay the pipeline to take the excess of gas, showing a negative US$9 in April, which contributed to an average price of only 73 cents per million Btu for the first eight months of 2019, compared with an average market price of US$2.10 in 2018 (which is also lower than the five year average from 2014 to 2018 of US$2.80) [17]. These trends create an opportunity for the development of technologies to transform shale/natural gas into value‐added chemicals. One additional point to consider is the increasing amount of liquefied natural gas that is being exported from the United States [18]. As this quantity grows, international natural gas prices may also get affected.
The main consumers of natural gas are the electricity generation industry, the residential sector, the industrial sector, and the chemical industry. Low natural gas prices have incentivized the electric power plants to switch from coal to natural gas, with an impact not only on the economy of these systems but also on the environment by reducing the total greenhouse gas emissions [1].
Another sector that has shown interest in switching from oil‐based feedstocks, such as naphtha or crude oil, to natural gas is the chemical industry. The availability of inexpensive natural gas and NGLs has boosted the chemical industry to create new plants for the production of value‐added chemicals using methane and NGLs as feedstock [5,19].
1.5 Alternatives to Produce Chemicals from Shale Gas
Due to the increasing availability of low‐cost natural gas, the chemical industry has started to invest in the research and development of chemical routes that can transform methane into value‐added chemicals. Some of the chemical compounds that have received special attention are methanol, ethylene, propylene, and liquid fuels obtained from syngas. Some of the processes to produce the aforementioned chemicals are discussed next.
Synthesis gas is a mixture of carbon monoxide and hydrogen typically needed for the production of chemicals such as a methanol, ammonia, or gas‐to‐liquid (GTL) products. The production process for synthesis gas varies depending on the oxidizing agent selected for the reforming of the natural gas. The main reforming processes are steam reforming (SR), partial oxidation (POX), and dry reforming (DR) [20]. The characteristics of these processes are listed in Table 1.3.
Table 1.3Reforming options and their characteristics.
Source: Adapted from Noureldin et al. 2014 [20].
| Reforming option |
Oxidizing agent |
Conditions |
Chemistry |
Type |
| Steam reforming |
H 2O |
Endothermic |
CH 4+ H 2O → CO + 3H 2 |
Catalytic |
| Partial oxidation |
O 2 |
Exothermic |
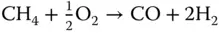 |
Catalytic/non catalytic |
| Dry reforming |
CO 2 |
Endothermic |
CH 4+ CO 2→ 2CO + 2H 2 |
Catalytic |
Although these processes may be used separately, combinations of two or more of the main reforming options have been proposed to enhance the overall performance of the reforming task. One such process is the autothermal reforming (ATR) in which the exothermic nature of the POX reforming is combined with the endothermic SR [21].
In all of these reforming alternatives, energy and water usage and generation are key points to consider when selecting the appropriate technology. Studies regarding heat and mass integration potential for the SR, POX, and ATR options can be consulted in the work of Martínez et al. [21] and Gabriel et al. [22].
Typically, methanol is used as an intermediate to produce other chemicals such as acetic acid, formaldehyde, and MTBE, among others [23]. The production process for methanol consists of three stages, reforming, synthesis, and purification. In the first stage, the main goal is to transform methane into syngas. For this purpose a reforming process is selected. One important factor to consider when selecting the reforming process is that the ratio of H 2to CO to feed the methanol synthesis reactor has to be equal to 2.
For the synthesis of methanol, compression of the syngas obtained from the reforming stage is needed. Then, the compressed syngas is fed to a catalytic reactor in which the following reactions take place:
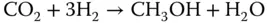
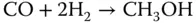
The synthesis reactor operates at 83 bar and 260 °C. The outlet of the reactor is cooled and sent to a flash unit to separate the unreacted syngas and recirculate it. Additionally, a fraction of the recycled syngas is purged, with a potential use as fuel. The crude methanol obtained from the flash unit is purified using one or two distillation columns [23].
This process has been analyzed to assess its environmental impact and its safety characteristics [23,24]. The main drawbacks of the process are the high pressure required for the operation of the synthesis reactor and the wasted fraction of non‐recycled syngas. Ortiz‐Espinoza et al. [24] studied the effect of different operating pressures for the methanol synthesis reactor on the safety, environmental, and economic characteristics of the methanol production process using POX reforming. The high operating pressure is related to the profitability of the process, but safety properties may be hindered by such operating conditions. Greenhouse emissions are an additional item of relevance for consideration. Figure 1.1shows the results of the analysis conducted by Ortiz‐Espinoza et al. [24], in which values of three metrics used for profitability, inherent safety, and sustainability are reported for different reactor pressures and recycling fractions for the unreacted syngas. Such metrics were the return on investment (ROI) for economic performance, process route index (PRI) for inherent safety, and total emissions of CO 2equivalents for process sustainability. One can observe the gradual trend of the three metrics that reflect their conflicting behavior. In summary, the economic potential of the process is better at high pressures and high recycling fractions, but if safety is of primary concern, a lower pressure would favor the process characteristics.
Читать дальше