While searching for new bio‐based resources for the synthesis of epoxy resins, particularly bisphenol substitutes, the crucial issue must be remembered. One of the most important challenges is to provide new bio‐based resins with comparable performance properties to the currently manufactured and applied petrochemical‐based commercial products, i.e. primarily high mechanical strength, thermal stability, and chemical resistance. The mentioned properties are characteristic of the resins based on bisphenol A (or other bisphenols), thanks to which these materials are produced on a large scale for many applications. Therefore, this chapter presents the most promising raw materials whose structure can provide the desired final properties of the epoxy system after cross‐linking. At the same time, they must be raw materials easily available in large quantities from renewable sources, nontoxic and cheap to obtain and in preparation.
1.2 Plant Oil Bio‐Based Epoxy Resins
Vegetable oils, as a material of natural origin and from renewable sources, are the subject of numerous studies aimed at their application for the synthesis or modification of various polymers [1]. Soybean, castor, linseed, rapeseed, sunflower, cotton, peanut, and palm oils are primarily used on a larger scale depending on the type of oil produced in a given region [2]. From the chemical point of view, plant‐based oils are a mixture of esters derived from glycerol and free fatty acids, mainly unsaturated acids (primarily oleic, linoleic, linolenic, ricinoleic, and erucic acids) and in a small amount of saturated acids (stearic and palmitic acids) ( Figure 1.1), depending on the type of oil.
When choosing vegetable oil for use in the synthesis of polymers, first of all, its structure should be taken into account: the presence of unsaturated bonds and possibly other functional groups (e.g. hydroxyl in castor oil or epoxy in vernonia oil), the amount of unsaturated bonds present in the molecule (referred as the oil functionality), and chain length alkyl derived from fatty acids ( Table 1.1).
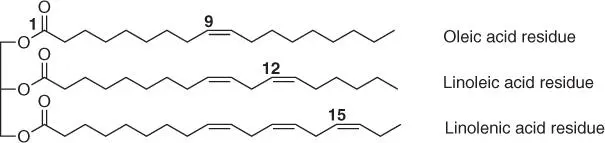
Figure 1.1 Schematic structure of triglycerides.
Table 1.1 The content of various fatty acids in selected vegetable oils.
| Fatty acid |
Vegetable oil, content of individual acids (wt%) |
| Soybean |
Rapeseed |
Linseed |
Sunflower |
Castor |
Palm |
| Palmitic |
12 |
4 |
5 |
6 |
1.5 |
39 |
| Stearic |
4 |
2 |
4 |
4 |
0.5 |
5 |
| Oleic |
24 |
56 |
22 |
42 |
5 |
45 |
| Linoleic |
53 |
26 |
17 |
47 |
4 |
9 |
| Linolenic |
7 |
10 |
52 |
1 |
0.5 |
— |
| Castor |
— |
— |
— |
— |
87.5 |
— |
| Other |
— |
2 |
— |
— |
— |
2 |
| Functionality |
4.6 |
3.8 |
6.6 |
4.6 |
2.8 |
1.8 |
The functionality of oils (understood as the content of unsaturated bonds) primarily determines the cross‐linking density of oil‐based chemosetting polymers or polymers obtained by free radical polymerization as well as oil‐modified polymeric materials. In turn, the final polymer properties such as mechanical strength, thermal stability, and chemical resistance strongly depend on the cross‐linking density. The elasticity of the polymers with the addition of vegetable oil or based on them depends on the length of the alkyl chains in the oil molecule derived from fatty acids.
Vegetable oils can be easily and efficiently converted into epoxy derivatives by oxidizing unsaturated bonds present in fatty acid residues. Several methods of double bond oxidation in triglyceride molecules are known and commonly used [3]: the method based on the Prilezhaev reaction, the radical oxidation, the Wacker‐type oxidation, dihydroxylation of oils and fats, and enzyme–catalyst oxidation. The Prilezhaev reaction is the most often used method for natural oil epoxidation, commonly applied in the industry. In this method, the process of epoxidation of natural fatty acids and triglycerols is carried out in the system consisting of hydrogen peroxide, an aliphatic carboxylic acid, and an acidic catalyst. The organic peracid formed in situ by the reaction of acid with hydrogen peroxide is the real oxidizing agent in this method ( Figure 1.2).
Carboxylic acids with one to seven carbon atoms are the most commonly used (in practice, mainly acetic acid). Inorganic or organic acids and their salts, as well as acidic esters, can be used as catalysts; however, sulfuric and phosphoric acid are the most often used in industrial practice. A promising method is oxidation in the presence of enzymes [4], heteropolyacids [5], and even ion exchange resins [6] as catalysts. The most commonly used oxidizing agent is hydrogen peroxide in the form of solution with a concentration of 35–90% (usually 50%). Epoxidation of plant oils in ionic liquids, as well as in supercritical carbon dioxide, is also described [7].
The earliest epoxidized esters of higher fatty acids have found wide applications as both plasticizers and stabilizers for thermoplastics, mostly poly(vinyl chloride), poly(vinylidene chloride), their copolymers, and poly(vinyl acetate) and chlorinated rubber [8, 9]. Epoxidized fatty acids containing oleic acid are used as a valuable intermediate in the production of lubricants and textile oils [10, 11]. It seems that epoxidized vegetable oils could also be used as hydraulic liquids [12]. However, primarily, they can also act as reactive diluents of bisphenol‐based epoxy resins [13], which are usually highly viscous. They have oxirane groups, although less reactive because of their central location in triglyceride chains (compared to terminal glycidyl groups), but also capable of reacting with polyamines or carboxylic anhydrides. By building into the structure of the cured resin in the process of co‐cross‐linking with it, they affect its final properties – improving flexibility and impact strength. In this way, embedded triglycerides not only facilitate the processing of resins with high intrinsic viscosity but also allow limiting their typical disadvantages (high brittleness, low impact strength, and flexibility) resulting from the rigid structure they owe because of the structure of bisphenols [14].
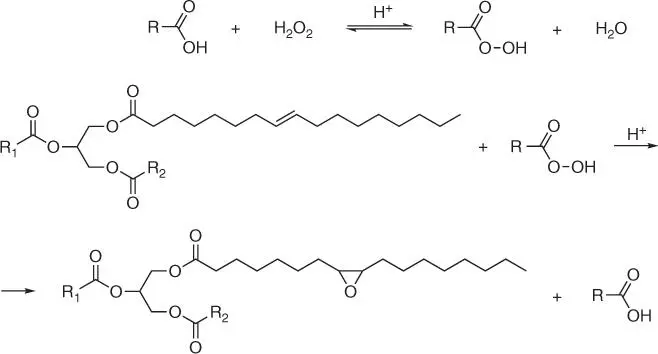
Figure 1.2 The reaction of triglyceride epoxidation with organic peracids.
However, the first, logically implied possibility of use of epoxidized vegetable oils is their application as stand‐alone materials: the networks cross‐linked with bifunctional compounds such as dicarboxylic acids or aliphatic and aromatic diamines, which are typically used as hardeners for the epoxy resins. Because of the content of more than one epoxy group in the molecule, epoxidized triglycerides may, according to the generally accepted definition, be treated as epoxy resins. However, curing of, e.g. epoxidized soybean oil [15] or vernonia oil (natural epoxidized oil mainly obtained from plant Vernonia galamensis ), dicarboxylic acids [16] resulted in obtaining only soft elastomers. Materials with higher mechanical strength are synthesized by reacting epoxidized oils first with polyhydric alcohols (e.g. resorcinol) and phenols or bisphenols and then cross‐linking the obtained modified oil with partially reacted epoxidized rings [17]. Finally, curing by photopolymerization or polymerization with latent initiators allows to obtain from modified vegetable oils, without the addition of the bisphenol‐based or cycloaliphatic resins, coating materials with satisfactory mechanical properties. It was found [18] that the properties of hardened vegetable oils also depend on the type of used thermal latent initiator. The properties of epoxidized castor oil cross‐linked with N ‐benzylpyrazine (BPH) and N ‐benzylquinoxaline (BQH) were studied ( Figure 1.3).
Читать дальше














