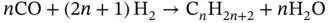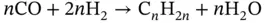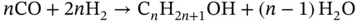Different materials and configurations can be used for the electrolyte and electrodes in solid oxide electrolyzers. The selection of the most suitable material will depend on the operation conditions, such as temperature or gas atmosphere. The type of electrolyte selected (protonic or ionic conductor) will set the reactions in each electrode. All electrolytes must possess some characteristics to ensure a good performance [108]. The electrolyte must be chemically, morphologically, and dimensionally stable in both atmosphere of the cell (oxidizing and reducing) and for all range of operation conditions. In addition, in order to minimize the ohmic losses in the cell performance, the electrolyte should have a good ionic or protonic conductivity in the cell operation conditions. The electronic conductivity should be as small as possible to avoid electron leakage in the electrolyte and the resulting low Faraday efficiency. Likewise, the porosity in the electrolyte has to be negligible to avoid gas leakage in the cell and consequently low performance. Finally, thermal expansion coefficient (TEC) should match with the adjacent components of the cell to avoid problems such as cracks and delamination. Moreover, TEC should be unalterable with oxygen partial pressure and temperature changes.
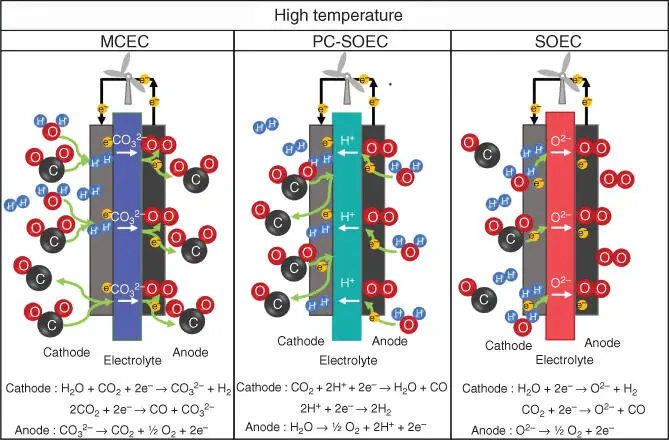
Figure 3.7Schematic representation of the CO 2and Co‐electrolyzer systems.
Regarding electrodes, some features have to be accomplished as well. Both, the oxidant and fuel electrodes have to be chemically, morphologically, and dimensionally stable in the working atmospheres and temperatures. To improve cell performance, the electronic conductivity has to be as large as possible. Additionally, oxygen ion or proton conductivity is required to extend the triple phase boundary (the point where electrons, gases, and ions are in contact and the electrochemical reaction takes place) along the whole electrode surface. Electrodes should have enough porosity to allow fast gas transport from/to the active reaction sites. TEC of electrodes should match the electrolyte and adjacent components along the operation conditions. Furthermore, electrodes have to match with other components in the operation and fabrication conditions. Finally, electrodes must exhibit enough catalytic activity (low polarization) for the different reactions that take place in the active sites, such as oxygen reduction reaction, hydrogen oxidation reaction, water splitting reaction, CO 2reduction, etc.
Other characteristics are also desirable for all components of the cell as the low cost, low toxicity, and manufacturability.
In the past years, several studies about PCEC have been published. Steam and CO 2co‐electrolysis was performed by Ruiz‐Trejo and Irvine using BaCe 0.5Zr 0.3Y 0.16Zn 0.04O 3−δaround 500 °C, obtaining promising results [109, 110]. Recently, Bausá et. al. reported co‐electrolysis at 700 °C using a BaCe 0.2Zr 0.7Y 0.1O 3−δelectrolyte [111].
3.5.2 Synthesis Gas Chemistry
Electrolysis consists in the dissociation of H 2O and/or CO 2by means of electricity. One of the main advantages of the co‐electrolysis is the production of H 2and CO simultaneously (synthesis gas or syngas). Syngas is widely used to produce a wide range of chemicals [112, 113]. The Fischer–Tropsch process is one of the main processes where syngas is involved. The Fischer–Tropsch process consists of the H 2and CO transformation into hydrocarbons of longer chains than methane. Hydrocarbons are the basis for the production of gasoline, diesel, and chemicals such as olefins and waxes. The type of catalyst selected (usually Fe and Co), design of the reactor, and the process conditions will shape the product selectivity in the Fischer–Tropsch process. Usually, Fischer–Tropsch synthesis takes place at temperatures between 200 and 300 °C and pressures comprised between 1 and 6 MPa. Among all the reactions that occur during Fischer–Tropsch synthesis, the main desired reactions can be addressed with the production of alkanes or paraffins (see Eq. (3.12)), alkenes or olefins (Eq. (3.13)), and alcohols (Eq. (1.14)) [114].
(3.12) 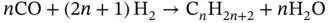
(3.13) 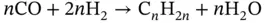
(3.14) 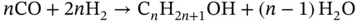
Apart from the reactions described above, some undesired reactions might also occur in the process as WGSR, carbonaceous materials, Boudouard reaction, and bulk carbide formation.
The synthesis of methanol and methane can also be derived from the syngas [115]. Methanation and methanol can be performed directly from the syngas or by CO 2hydrogenation. The main reactions for both syntheses are described below. For both processes, WGSR plays an important role and affects the selectivity to the final products.
1 (a) CO2 + 4H2 → CH4 + 2H2O
2 (b) CO + 3H2 → CH4 + H2O
3 (c) CO + H2O → CO2 + H2
1 (a) CO2 + 3H2 → CH3OH + H2O
2 (b) CO + 2H2 → CH3OH
3 (c) CO + H2O → CO2 + H2
Furthermore, products obtained from the Fischer–Tropsch reaction and methanol can also be recombined with syngas, CO, or H 2to obtain different products highly valued in the chemical industry. Some examples for different products are, for instance, ethanol to gasoline reaction by means of zeolites [116, 117]; hydroformylation reaction of olefins with syngas to aldehydes, and alcohols with cobalt and rhodium catalysts [118]; carbonylation reactions [119], where CO from the electrolysis can be employed; and methanol plus syngas or carbon monoxide, for the synthesis of value‐added chemicals [117].
The important role of electrolysis as a bridge between renewable energies, energy storage technology, and value‐added products (chemicals, fuels, etc.) is therefore obvious. The combination in the same step of both technologies, CO 2co‐electrolysis to produce syngas and the production of hydrocarbons by Fischer–Tropsch process, will give rise to a more efficient, compact, and environmental friendly technology. The eCOCO 2project [120] (sponsored by the EU commission via the H2020 program) based on CO 2conversion focuses on the development of co‐ionic electrochemical cells, which enable both: the electrolysis of water and the hydrocarbon synthesis in the same step. The CO 2converter consists of electrochemical cell constructed with a co‐ionic electrolyte that allows the injection of protons to the reaction cell, and the simultaneous extraction of oxygen ions. In addition, a multifunctional catalyst will be integrated in the electrochemical cell for the hydrocarbon generation. The final objective of this project is the production of more than 250 g of jet fuel per day.
3.5.3 Other Applications
3.5.3.1 Methane Steam Reforming
The feasibility of performing SMR using the proton‐conducting material BaZr 0.7Ce 0.2Y 0.1O 2.9(BZCY72) was successfully examined at low temperatures (450–650 °C) under atmospheric pressure by M. Stoukides and coworkers [121]. The system exhibited strong dependence on gas concentration, temperature, and applied voltage, as well as excellent chemical stability.
Читать дальше