Capture, purification, and transport of CO 2are essential for its utilization. Table 1.3lists several large CO 2sources with their typical amounts and concentrations of CO 2as well as impurities. In present CO 2merchant market (approximately 230 Mton, US$7.7 billion), the fermentation process (i.e. bioethanol production) and ammonia production, which provide close to 100% CO 2, are predominantly CO 2sources [5, 30]. The CO 2generated from ethanol fermentation commercially supplies roughly 270 000 ton of CO 2annually for EOR through pipeline from Kansas to Texas [28]. On the other hand, the production of electricity and heat accounts for 41% of global CO 2emissions ( Figure 1.4), and the transport and industrial sectors account for an additional 25% and 19%, respectively [32]. However, suitable sources of CO 2for use in chemical transformation are limited. The gases contain various impurities, the separation of which is both energy and cost intensive. To supply CO 2of an appropriate quality for use in chemical conversion processes, capture and separation are required ( Table 1.4) [33]. The most effective CO 2capture method as the current industrial standard is chemical absorption in an aqueous solution of an amine‐based organic compound. However, the cost (35 US$/ton) and energy consumption (2.5 GJ ton −1) of amine capture must still be reduced to provide economically viable routes from carbon dioxide to fuels [34, 35].
Table 1.3Concentration of CO 2and contaminants from various sources.
Source: Carbon Recycling International; Capturing and Utilizing CO 2from Ethanol: Adding Economic Value and Jobs to Rural Economies and Communities While Reducing Emissions (2017); and Greenhouse Gas Inventory Data [9, 28, 29].
| Source |
Amount/Mton |
CO 2concentration/% |
Impurities |
| Ethanol fermentation [28, 30] |
50 |
99 |
EtOH, MeOH, H 2O, H 2S |
| Anhydrous ammonia |
30 |
>95 |
NH 3, CO, H 2, H 2O |
| Natural deposits |
13 |
90–100 |
N 2, O 2, He |
| Power plants |
4287 |
10–15 |
N 2, H 2O, SO x, NO x, CO |
| Steelmaking |
266 |
18–20 |
N 2, SO x, NO x, O 2 |
| Cement production [31] |
220 |
14–33 |
SO x, NO x, O 2 |
| Atmosphere |
3 200 000 |
0.04 |
N 2, O 2, SO x, NO x |
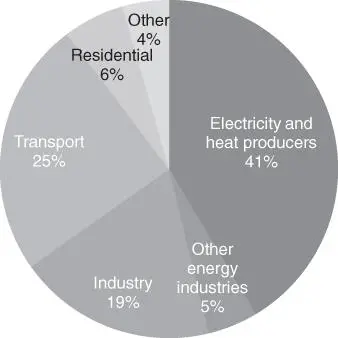
Figure 1.4CO 2emissions from fuel combustion.
Source: Data from IEA, CO 2emissions from fuel combustion, 2020 [32].
Table 1.4CO 2capture technologies.
Source: Based on Styring [33].
| Capture technology |
Technical principle |
| Chemical absorption |
Chemical reaction between CO 2and absorbent by a temperature swing. |
| Physical absorption |
Dissolution of CO 2into a liquid, the efficiency of which depends on the solubility of CO 2in the liquid. |
| Solid absorption |
Absorption into solid absorbents, which include porous materials impregnated with amines for low‐temperature separation or other solid absorbents for high‐temperature separation. |
| Physical adsorption |
Adsorption onto porous solids such as zeolites by a pressure or temperature swing. |
| Membrane separation |
Permeation through a membrane with selective permeability for different gas species. |
Recently, the direct capture of CO 2from ambient air, called direct air capture (DAC), has received increasing attention [36]. One of the advantages of DAC is that it can be located anywhere, because it is unnecessary for CO 2transport. However, from both engineering and chemistry views, there remains much room for improvements to the sorbents and processes. Additionally, thorough techno‐economic analyses of DAC processes are necessary [37].
1.4.2 Energy and H 2Supply
Another consideration is the energy required to capture and convert CO 2, which must certainly be derived from renewable sources ( Figure 1.5) [38]. If this energy comes from fossil oils, much more CO 2will be emitted than separated. Fortunately, the renewables now account for over 25% of global power output (hydro: 16%, wind: 5%, PV: 2%), [1] and the costs of PV and wind power become even lower than that of fossil fuels (natural gas and coal) ( Figure 1.6) [39]. Thus, electricity from renewable sources can be converted into H 2by water electrolysis, which can be performed on an industrial scale. Nevertheless, H 2produced by electrolysis systems (2.5–6 US$/kg H2) is at present more expensive than that from current industrial production based on conventional fossil sources, like natural gas reforming and coal gasification (<1 US$/kg H 2) [40, 41].
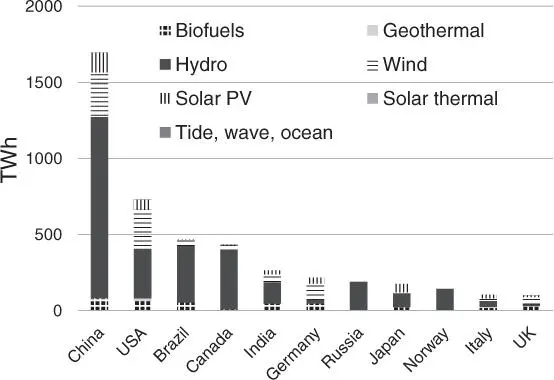
Figure 1.5Low‐carbon electricity generation by source in 2017.
Source: Data from explore energy data by category, indicator, country or region (IEA) [38].
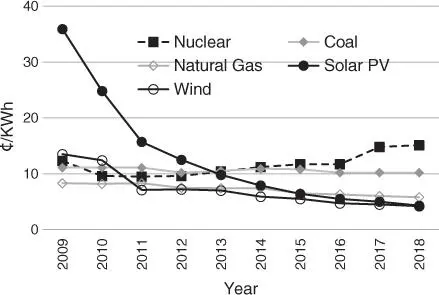
Figure 1.6Levelized cost of energy comparison: Renewable energy versus conventional generation.
Source: Data from Lazard.com, Lazard's levelized cost of energy analysis [39].
1.5 Political Aspect: Tax
The future prospects for CO 2utilization on large scale will mainly depend on policy support. The carbon tax, a fee imposed on the burning of carbon‐based fuels (coal, oil, gasoline, and natural gas), is one policy for reducing the use of fossil fuels. To reduce CO 2emissions, as many as 29 countries have implemented carbon taxes as of 2019. Tax rates, including energy taxes, differed according to use and fuel type in 2017. For example, high tax rates are imposed on gasoline in every country, from  in the United States to
in the United States to 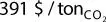 in the Netherlands [42]. On the other hand, there are also significant differences in tax rates for the industrial sector depending on the country ( Figure 1.7). An increasing of the tax rates of carbon‐based fuels seems to be necessary to motivate our societies to switch to clean energy.
in the Netherlands [42]. On the other hand, there are also significant differences in tax rates for the industrial sector depending on the country ( Figure 1.7). An increasing of the tax rates of carbon‐based fuels seems to be necessary to motivate our societies to switch to clean energy.
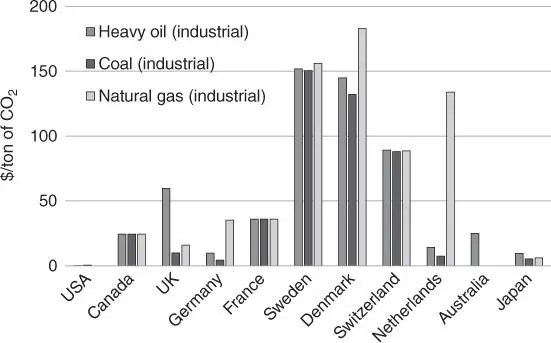
Figure 1.7Carbon tax rate per ton of emitted CO 2.
Source: Data from the Ministry of the Environment of Japan [42].
1.6 Conclusion and Perspectives
CO 2utilization will play a crucial role in achieving the internationally agreed climate and energy goals. In particular, the conversion of CO 2to fuels and chemicals will be of significant importance. However, these technologies are still in their infancy, and the following issues require consideration and technological improvements:
1 (i) Supply of H2 and power from renewables.
2 (ii) Cost reduction, mainly for the supply of low‐carbon H2.
3 (iii) Political support to shift from a fossil‐based to CO2‐based economy.
Читать дальше




 in the United States to
in the United States to 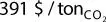 in the Netherlands [42]. On the other hand, there are also significant differences in tax rates for the industrial sector depending on the country ( Figure 1.7). An increasing of the tax rates of carbon‐based fuels seems to be necessary to motivate our societies to switch to clean energy.
in the Netherlands [42]. On the other hand, there are also significant differences in tax rates for the industrial sector depending on the country ( Figure 1.7). An increasing of the tax rates of carbon‐based fuels seems to be necessary to motivate our societies to switch to clean energy.
