Notably, the amount of CO 2utilized by all these approaches, including urea and carbonate production, is very small compared with the magnitude of anthropogenic emissions. Therefore, CO 2conversion into chemicals is unlikely to significantly reduce emissions. Comparatively, it should be noted that fuels are produced and consumed on a much larger scale than these chemicals.
1.3 Application and Market Studies of CO 2Hydrogenation Products
Hydrogenation of CO 2could be an efficient option for developing more environment‐friendly products as alternatives to fossil‐based ones. In terms of practicality, the distribution infrastructure of carbon‐based chemicals is well established. However, their manufacturing is currently several times more expensive than their conventionally produced counterparts, mainly due to the costs associated with H 2production. Some of the key features of CO 2hydrogenation products and conventional fuels are given in Table 1.2.
Table 1.2Characteristics of various energy vectors.
| Compound |
Energy density (GJ m −3) |
Approx. price per energy (US$/GJ) |
Boiling point/melting point (°C) |
Ignition point/flash point (°C) |
Vapor pressure at 25 °C (kPa) |
| Methanol |
15.8 |
15 |
64.55/–97.68 |
470/15 (open) |
16.9 |
| Formic acid |
6.3 |
100 |
100.56/8.27 |
520/59 (open) |
43.1 |
| Natural gas (CH 4) |
8.1 (20 MPa) |
2 |
–161/–183 |
537/–188 |
147 (15 °C) |
| Gasoline |
34.5 |
30 |
17–220/≤–40 |
300/≤–43 |
50–93 (37.8 °C) |
| Diesel oil |
36.3 |
23 |
140–400/–29 to –18 |
250/40–70 |
≤0.35 (37.8 °C) |
| Hydrogen |
5.1 (70 MPa) |
120 |
–252.87/–259.14 |
500–571/— |
1.65 × 10 5 |
1.3.1 Formic Acid/Formate
Formic acid is the first carboxylic acid and is naturally occurring produced by ants, bees, and some plants. In 2016, the global production of formic acid was 1.02 Mton [10]. The general production process of formic acid involves the formal carbonylation of water in a two‐step synthesis via methyl formate. Formic acid and its salts (formate) are valuable chemical products used for silage and animal feed (27%), leather and tanning (22%), pharmaceuticals and food chemicals (14%), textile (9%), natural rubber (7%), and drilling fluids (4%) [11]. Recently, formic acid has been recognized as a promising liquid organic H 2carrier (LOHC) because of its low toxicity, low combustibility, stability, environmental friendliness, and 4.4 wt% (53 g l −1) H 2content [12–14]. In addition, compressed hydrogen gas can be supplied only by heating of formic acid using catalysts as a chemical compressor [15]. Therefore, advances in the efficient production of formic acid/formates may eventually lead to their large‐scale use as LOHCs (see Chapter 10).
Methanol, the industrial production of which is mainly from syngas, is in high global demand as a fuel and bulk chemical ( Figure 1.3) [17]. One ton of methanol produced by the established process consumes 37.5 GJ of natural gas and emits 1.49 ton of CO 2[18]. In 2018, the global production of methanol was approximately 91.7 Mton, and since 2015, its production has grown by approximately 16% [16]. Approximately 26% and 8% of the methanol produced worldwide is consumed to produce formaldehyde and acetic acid, respectively, as the conventional demands. Methanol can be used as a fuel for internal combustion engines and fuel cells because it has a comparably high‐octane number of 113 and a density approximately half that of gasoline. In addition, methanol can be transformed into gasoline through the methanol‐to‐gasoline (MTG) process developed by Mobil in the 1970s [19]. Another growing market for methanol is the production of light olefins (i.e. ethylene (152 Mton yr −1) and propylene (103 Mton yr −1) in 2017), [20] which are monomer feedstocks for polyethylene and polypropylene as basic products of the plastics industry [21]. The concept of a so‐called methanol economy was independently proposed by Olah and Asinger due to the chemical's promising characteristics for use as an energy vector and chemical feedstock [22–24]. Therefore, the production of methanol by CO 2conversion is regarded as an attractive and potentially profitable route for CO 2utilization.
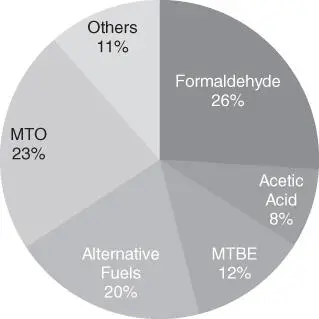
Figure 1.3Global methanol demand in 2018.
Source: Data from Global methanol demand (Methanol Institute) [16].
CO 2methanation, also known as the Sabatier process, affords methane by the exothermic reaction of CO 2with H 2. The commercial methanation of CO 2is performed at 300–550 °C and above 5 bar. Most CO 2methanation processes are considered to be a linear combination of rWGS and CO methanation. The process is expected to be a power‐to‐gas concept for converting renewable electrical energy into methane as chemical energy. In other words, the main goal of methanation is the intermediate storage of renewable electricity in methane as an energy carrier. Since fossil‐based natural gas is a common fuel, there would be easy access to existing infrastructure.
Due to the significant interest in CO 2methanation, the first pilot plant capable of producing 0.5 Nm 3h −1of synthetic natural gas was built in Japan [25]. In terms of commercial installations, Audi has an operational CO 2methanation facility (max. 325 Nm 3h −1) using renewable H 2(max. 1300 Nm 3h −1) from electrolysis (max. 6.0 MW) in Germany [26, 27].
The two most growing renewable energy sources, solar and wind, are intermittent and thus provide highly fluctuating electrical energy. In addition, the region's best suited areas for the production of renewable energy are often far from consumption areas, i.e. cities. These cause the two key problems of storage and transport. Certainly, electrical energy is an effective way to transfer energy within 1000 km and can be stored in batteries. However, low‐cost solutions for the large‐scale storage and long‐range transport of electrical energy must be developed to improve energy security and balance energy prices.
The transformation of excess renewable energy into chemical energy by converting CO 2is one promising option. CO 2‐based compounds, such as methane, methanol and formic acid, can store energy as gas or liquids with comparably high‐energy densities. Especially liquids can be easily transported and release energy as H 2or electricity through oxidation and fuel cells when there is a greater demand. In other words, CO 2can act as an energy vector between electrical and chemical energy. Recently, the electroreduction of CO 2to chemical fuels has been receiving increasing attention because it allows for the direct use of renewable electricity without conversion to high‐cost H 2by water electrolysis (see Chapter 9). Much more CO 2is in demand as a feedstock for fuels than for chemicals and mineralization. In addition, related photo‐catalytic processes gain more and more interest.
The CO 2hydrogenation approach requires H 2, CO 2, and an energy supply. In particular, how much the energy‐intensive hydrogenation process contributes to mitigating CO 2emissions will be dominated by the H 2source. Obviously, H 2must be produced with the help of a renewable electricity source such as water electrolysis and not from fossil fuels.
Читать дальше


