ePub ISBN:978‐3‐527‐82410‐6
oBook ISBN:978‐3‐527‐82411‐3
Carbon dioxide is widely considered to be primarily responsible for global climatic changes. Presently, scientists are facing enormous challenges in mitigating the global CO 2emissions. Significant progress has recently been achieved in the research topic of the catalysis of CO 2hydrogenation, as one of the most important subjects in chemistry. In addition, the paradigm shift from fossil fuels to low‐carbon renewable energy (solar photovoltaics and wind) in recent years will allow for the competition between the CO 2emission by energy consumption and its fixation by CO 2conversion. In future, advancement in the fields of carbon capture and utilization is expected.
I would like to thank all the authors, who are all acknowledged as world expert in their area of CO 2hydrogenation, for their enthusiastic efforts to present recent advances in CO 2hydrogenation. Their state‐of‐the‐art research gives exceptionally beneficial information to the researchers, teachers, and students who are interested in the research field of CO 2hydrogenation. I anticipate that their contributions will stimulate further study in CO 2utilization as well as CO 2hydrogenation. I would also like to thank the Wiley‐VCH team for their continuous support. Finally, I deeply appreciate the members of my research group for their valuable assistance, especially Dr. Ryoichi Kanega for the cover design, and Dr. Hide Kambayashi for data survey.
In the spring and summer of 2020, the world has been hit by the COVID‐19 pandemic. Despite these difficult times, I am delighted that this book could be completed.
July 2020
Yuichiro Himeda
National Institute of Advanced Industrial Science and Technology,
Global Zero Emission Research Center,
Tsukuba, Japan
Yuichiro Himeda 1 and Matthias Beller 2
1National Institute of Advanced Industrial Science and Technology, Global Zero Emission Research Center, AIST Tsukuba West, 16‐1 Onogawa , Tsukuba , Ibaraki, 305‐8569 , Japan
2Leibniz‐Institut für Katalyse, Applied Homogeneous Catalysis, Albert‐Einstein Straße 29a , 18059 , Rostock, Germany
Of the final products of the combustion of carbon‐based fossil fuels, carbon dioxide (CO 2) has the highest oxidation state and is known as the major cause of global warming. Annual CO 2emissions from anthropogenic activity in 2018 were approximately 33.1 Gton, an increase of 1.7% compared with 2017 [1]. Since the Industrial Revolution, two trillion tons of CO 2have accumulated in the atmosphere, and the current atmospheric concentration of CO 2has reached an unprecedented level of over 400 ppm ( Figure 1.1) [2]. The anthropogenic emission of CO 2is associated with energy consumption, i.e. the combustion of carbon‐based fossil fuels, which currently account for around 85% of the world's energy.
According to the Paris Agreement of the United Nations, an overall limit on total cumulative CO 2emissions is crucial for our future development [3, 4]. According to the 2 °C scenario, further cumulative emissions should be limited to below one trillion ton of CO 2. The spread of renewable energy (35%), advances in energy conservation (40%), and carbon capture and sequestration (CCS) technologies (14%) are sure to contribute to addressing the problem ( Figure 1.2) [3]. However, it is clear that these methods will not completely solve the issues arising from the vast quantities of emitted CO 2. In 2017, the International Energy Agency (IEA) presented the Energy Technology Perspectives (Beyond 2 °C Scenario: B2DS), which placed a much greater emphasis on the role of CO 2utilization for reducing emissions [3]. Indeed, in the next decade, we will still rely on carbon‐based products for fuels, polymers, commodity chemicals, cosmetics, detergents, and fabrics in modern life. If these chemicals were to be derived from CO 2instead of fossil oils, a sustainable carbon cycle will be possible.
Apart from chemical applications, already today, CO 2is used directly in enhanced oil recovery (EOR), beverage carbonation, food processing (e.g. coffee decaffeination and drinking water abstraction), welding, as a cleaning agent for textiles, and as a solvent in the electronics industry [5]. These approaches are commercially viable. In particular, 70–80 Mton of CO 2is consumed for EOR in the oil sector. Although such direct utilization of CO 2addresses a significant amount of CO 2emissions, these topics are beyond the scope of this book.
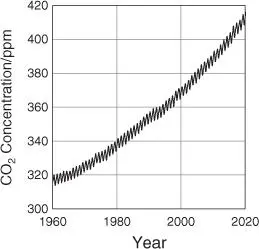
Figure 1.1Atmospheric CO 2concentration at Mauna Loa Observatory.
Source: Data from National Oceanic and Atmospheric Administration, Global Monitoring Laboratory [2].
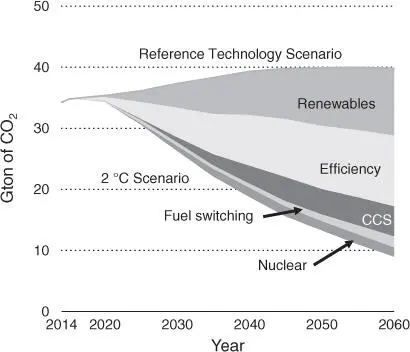
Figure 1.2IEA 2 °C Scenario (2DS) in Energy Technology Perspectives 2017.
Source: Data from Market‐driven future potential of Bio‐CC(U)S [3].
1.2 Chemicals from CO 2as a Feedstock
CO 2has been recognized as an inexpensive and abundant industrial C1 carbon source. The various chemicals that can be produced by CO 2conversion are shown in Table 1.1[6]. The largest chemical use of CO 2is in the production of urea from ammonia. However, since a huge amount of CO 2is emitted during methane steam reforming to supply H 2, urea production does not contribute to carbon sequestration at present.
Table 1.1Chemicals produced commercially from CO 2.
Source: From Omae [6]. © 2012 Elsevier.
| Chemical |
Scale of production/ton |
| Anthropogenic CO 2emissions (2018) |
33 100 000 000 |
| Urea [7] |
181 000 000 |
| Diphenyl carbonate (Asahi Kasei Process) [8] |
1 070 000 |
| Salicylic acid |
90 000 |
| Cyclic carbonate |
80 000 |
| Polypropylene carbonate |
76 000 |
| Acetylsalicylic acid |
16 000 |
| Methanol (CRI process) [9] |
4000 |
The catalytic copolymerization of CO 2with epoxides, which provides a thermodynamic driving force due to the strained three‐membered ring, is the most prominent example of the synthesis of CO 2‐based polymers without formal reduction of the carbon oxidation state. Another example, the manufacture of diphenyl carbonate from ethylene oxide, bisphenol A, and CO 2instead of phosgene was developed beginning in 1977 by Asahi Kasei Chemical Corporation to address environmental and safety issues. The first commercial facility started operation in 2002 [8]. This process produces high‐quality polycarbonate and high‐purity monoethylene glycol in high yields without waste or wastewater. In addition, the phosgene‐free process emits approximately 2.32 ton/ton PCless CO 2than the phosgene process according to life‐cycle assessment (LCA). Diphenyl carbonate has a large market (3.6 Mton in 2016) for use in automotive parts and accessories, glazing, and medical devices. The phosgene‐free technology has already been licensed to Taiwan, South Korea, Saudi Arabia, China, and Russia.
Since 2011, in Iceland, carbon recycling international (CRI) operated the first commercial plant for methanol production from CO 2via syngas by the reverse water‐gas shift (rWGS) reaction (George Olah Renewable Methanol Plant) [9]. At present, more than five million liters of methanol per year is produced using low‐cost electricity and high‐concentration CO 2in the flue gas from an adjacent geothermal power plant. It should be noted that this technology is at present only viable in Iceland; however, if there is a surplus of green electricity in the future from an excess of renewable energy, then this process will be attractive at other places, too.
Читать дальше



