Recently, it has been suggested that platelet and MNC donors may experience prolonged lymphopenia after repeated donations [124]. It is unclear whether this effect is instrument dependent and/or related to other factors such as donor age, and studies are ongoing. Thus far, there appear to be no reports of medical complications associated with this apparent lymphopenia.
Characteristics of the peripheral blood stem cell concentrates
Large quantities of CD34+ cells can be collected from normal donors given G‐CSF for 5 days. G‐CSF doses of 7.5 or 10 mg/day provide a greater yield than 5 mg/day [115], but it is not clear whether there are statistically significant differences in the CD34+ cell yield between 7.5 and 10 mg/day. Because the donor side effects increase with increasing doses of G‐CSF, we recommend that donors be given 7.5 mg/kg/day of G‐CSF to mobilize peripheral blood CD34+ cells [115]. The composition of the PBSC component is shown in Tables 6.3and 6.4. Most PBSC components contain only a small volume of red cells (mean, 7 mL) but a rather large total number of neutrophils and platelets [115]. This number of platelets is similar to the number of platelets ordinarily provided in a platelet transfusion, and if transfused fresh (i.e., allogeneic transplant) this may provide an additional benefit from the PBSC transfusion. The PBSC concentrate usually has a volume of about 200 mL and contains approximately 3 × 10 10MNCs and 4 × 10 8CD34+ cells.
Quality control of peripheral blood stem cell concentrates
Because there is no definitive test for the primordial hematopoietic stem cell, quality control of these PBSC concentrates is not standardized. Cell culture techniques can be used to determine CFU‐GM, BFU‐E, and CFU‐MIX colonies, and the number of CD34+ cells can be determined by flow cytometry. In practice, the dose for transplantation is usually based on cell counting to obtain at least 3 × 10 8mononuclear or 5 × 10 6CD34+ cells per kilogram of the recipient’s body weight. The results of progenitor assays are not available for about 2 weeks and thus can be used only in retrospect for research purposes. Many centers do not even determine progenitor content because they do not believe there is a correlation with engraftment, although we believe this can be a valuable quality‐control test.
Table 6.3 Effects of 10 μg/kg G‐CSF dose on the quantity of cells collected by one apheresis procedure from healthy people treated with G‐CSF for 5 days (number of cells collected).
Source : Stroncek DF, Clay ME, Smith J, et al. Composition of peripheral blood progenitor cell components collected from healthy donors. Transfusion 1997; 37:411–417. © 1997 John Wiley & Sons. Reproduced with permission of John Wiley & Sons.
|
All components ( n = 150) |
| Cell type |
Mean ± SD |
Median |
Range |
| WBCs (× 10 9) |
39.8 ± 21.8 |
36.0 |
15.6–163.3 |
| MNCs (× 10 9) |
38.1 ± 19.4 |
34.5 |
15.6–139.7 |
| CD34+ cells (× 10 8) |
452 ± 294 |
383 |
78–1,380 |
| CD34+ cells (× 10 6per L processed) |
53.2 ± 33.1 |
46.5 |
9.3–146.3 |
| RBC (mL) |
7.2 ± 3.5 |
7.6 |
0–15.5 |
| Neutrophils (× 10 9) |
1.77 ± 3.37 |
1.05 |
0–23.68 |
| Platelets (× 10 11) |
490 ± 100 |
490 |
250–740 |
MNC, mononuclear cell; RBC, red blood cell; WBC, white blood cell.
Table 6.4 Quantity of cells in the PBSC components.
Source : Stroncek DF, Clay ME, Smith J, et al. Comparison of two blood cell separators in collecting peripheral blood stem cell components. Transfus Med 1997; 7:95–99. © 1997 John Wiley & Sons. Reproduced with permission of John Wiley & Sons.
| Cell type |
CS‐3000 ( n = 15) |
Spectra ( n = 14) |
P |
| WBCs (× 10 9) |
40.9 ± 21.7 |
33.1 ± 10.7 |
0.24 |
| Neutrophils (× 10 9) |
1.38 ± 1.88 |
5.53 ± 8.71 |
0.001 |
| Mononuclear cells (× 10 9) |
39.6 ± 21.9 |
26.9 ± 5.6 |
0.02 |
| Platelets (× 10 9) |
507 ± 98 |
531 ± 116 |
0.54 |
| CD34+ cells (× 10 6) |
470 ± 353 |
419 ± 351 |
0.69 |
WBC, white blood cell.
There is considerable variation in the number of CD34+ cells collected ( Figure 6.3). In our early experience [115], a single‐cytapheresis procedure yielded a median dose of 780–1,658 × 10 6CD34+ cells. In approximately 42% of the procedures, this would be an adequate cell dose to transplant 5 × 10 6CD34+ cells/kg to a 75‐kg recipient.
In 86% of donors, two cytapheresis procedures would yield an adequate cell dose for transplanting the 75‐kg recipient. These numbers were obtained by processing approximately 10 L of whole blood, and most centers now process 15–20 L. Other reports involving processing of 15–20 L of blood for each cytapheresis procedure suggest that larger numbers of CD34+ cells are obtained [114]. Thus, currently for most donors, one or two procedures result in a dose of cells suitable for transplantation.
Storage of peripheral blood stem cells
Because of the variability in the number of cells that may be obtained, the strategy for using the cells for transplantation cannot always be the same. If the dose needed for transplantation can be obtained with one procedure, the cells can be transfused immediately. However, if two or three apheresis procedures are necessary, it may be desirable to freeze the concentrates and transfuse them all at once. However, the freezing and thawing may alter the composition of the PBSC concentrates, and so some transplant physicians give the cells fresh each day until the desired dose is obtained. Alternatively, the concentrate collected on the first day is stored in the liquid state and transfused with the concentrate collected on the second day. It appears that PBSCs can be preserved satisfactorily in Plasmalyte A, Normosol or STM‐Sav for 24 hours at room temperature [125]. A more extensive discussion of hematopoietic stem cell preservation is provided in Chapter 19.
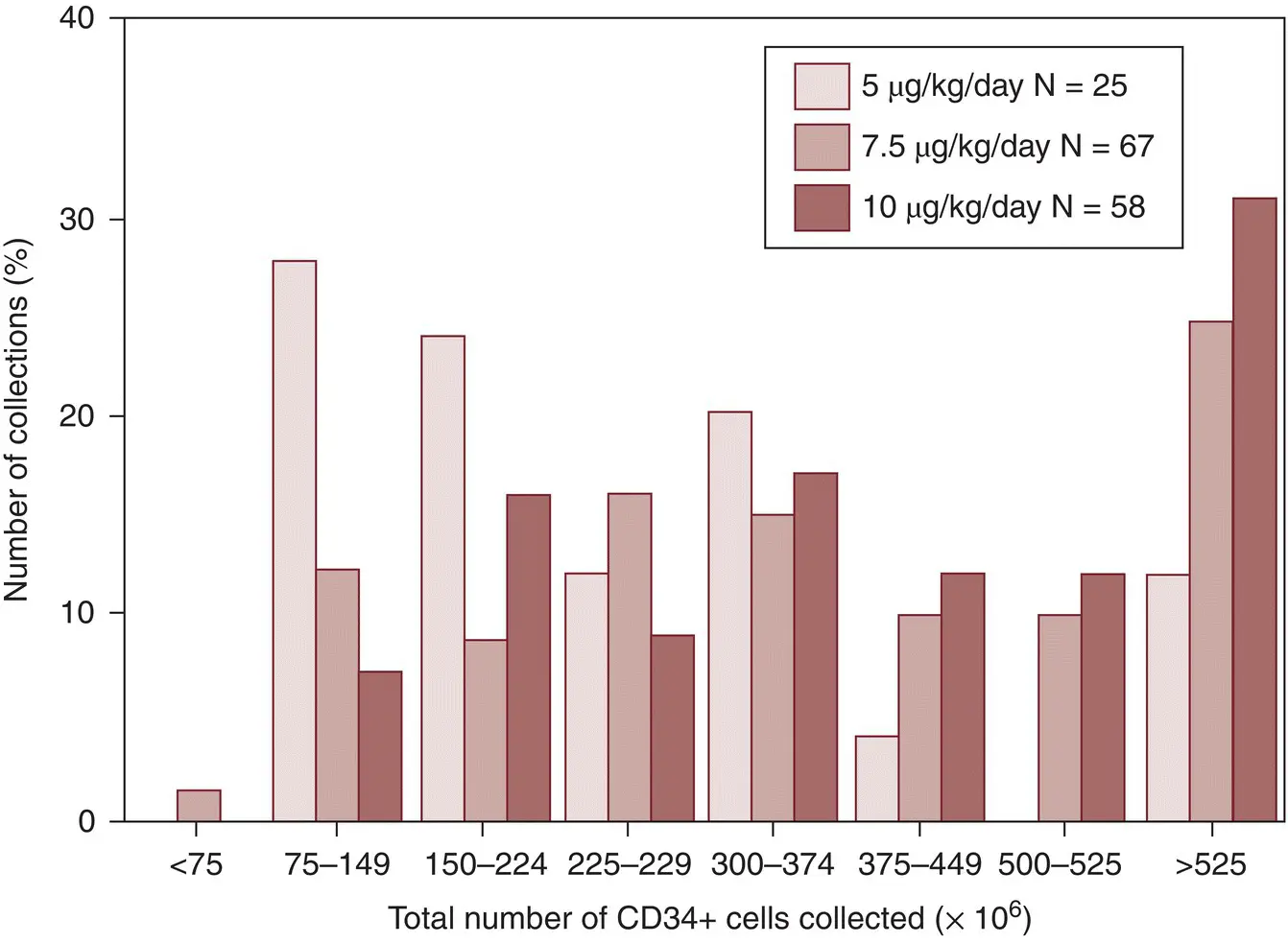
Figure 6.3 CD34+ cell yield in peripheral blood concentrates collected from normal donors.
( Source : Reproduced with permission from Stroncek DF, Clay ME, Smith J, et al. Composition of peripheral blood progenitor cell components collected from healthy donors. Transfusion 1997; 37:411–417. © 1997 John Wiley & Sons. Reproduced with permission of John Wiley & Sons.)
6.7 Donor selection and complications of cytapheresis in normal donors
Because donation of blood components by apheresis is fundamentally different from whole blood donation, there are some donor eligibility requirements and complications that are unique to apheresis donors. This chapter focuses on the donation procedures and the products. Donor selection and complications are discussed in Chapter 4.
6.8 Plasmapheresis and source plasma
The plasma collection and fractionation industry in the United States developed during the 1960s using manual plastic bag methods for plasma collection by plasmapheresis. Today, virtually all source plasma collected in the United States for fractionation into derivatives (see Chapters 2and 5) is obtained by semiautomated instrument plasmapheresis. It has been estimated [126] that about 28 million liters of plasma are fractionated annually in the world (see Chapter 2). Most plasma used as fresh frozen plasma (FFP) is obtained from whole blood, but the increasing flexibility of some apheresis instruments makes it possible to obtain plasma for FFP as a by‐product of platelet or red cell apheresis. There are no data on the number of plasma products produced in this manner. Apheresis plasma contains greater activities of factor V, factor VIII, factor IX, and factor XI, prothrombin fragments 1 and 2, and platelet factor IV compared with recovered plasma (see Chapter 4and Burnouf [126] and Runkel et al. [127]). Thus, apheresis appears to produce plasma with a higher quality, although the clinical significance of this is not established.
Читать дальше