In periodic table, mercury belongs to the transition element series. It occurs in three forms in the nature as elemental, inorganic, and organic form with individual toxicity character [43]. Generally, it exists in liquid form [23]. It can also exist as a cation through oxidation state of +1 (mercurous) or +2 (mercuric) [44]. It is used in electrical industry, dental amalgams, and in various industrial methods containing the manufacture of caustic soda, antifungal agent, manufacture of nuclear reactors, usage as a solvent for reactive and expensive metal, as a preservative of pharmaceutical products, etc. [45]. Mercury is released and contaminates the environment from combustion of coal, manometers at gas pressure measuring stations, and gas/oil pipelines as well. It exists in mercuric (Hg 2+), mercurous (Hg 2 +2), elemental (HgO), or alkylated (methyl/ethyl mercury) form [23]. Human and animals expose to mercury and other chemicals by calamities, atmospheric pollution, contamination of food, dental repair, precautionary medical applications, farming, and industrial processes [17, 30]. Mercury entered to water by natural procedure of gassing from ground of earth and by industrial pollutants and accumulates in fish and tiny organisms inhabiting in the water bodies. Due to its lipid soluble nature, it can easily cross placenta and blood brain barrier. By eating methyl mercury affected fish, it enters to gastrointestinal tract and also affects kidney, neurological tissue and liver of human and it causes gastrointestinal toxicity, neurotoxicity, and nephrotoxicity [16].
3.2.2 Essential Heavy Metals
The essential heavy metals such as nickel, zinc, and copper are require for biological metabolism but in high concentration they show toxic and harmful effects and on living organisms.
Copper (Cu) is a transition element, occupied a place in group IB of period 4 in periodic table and ranked as third highest used metal in the world [23, 46]. It is a fundamental micronutrient for animal and plant development. In human, it helps for production of blood hemoglobin and in plant, Cu is essential for seed germination, resistant to disease and water regulation [23]. Spontaneously high amount of exposure of cupper dust causes eye, nose, and mouth irritation and often causes nausea and diarrhea [22]. Continuously exposure to high doses of Cu causes anemia, malfunction of liver, kidney, and impatience in stomach and intestine [23]. Generally, copper (Cu) is present in drinking water because flow pipes are made up of copper to control algal growth [23]. Mining, metallurgy, and industrial applications are the important causes of copper contamination in the environment [22].
Zinc occupied a place in group IIB of periodic table. It is a transition element normally present in soil approximately 70 mg kg −1in ground rocks [47]. It is an essential micro nutrient in our diet but higher concentration is toxic and may cause anemia and cholesterol complications in human beings and nausea and vomiting in children [22]. Currently, Zn concentration is found to be rising because of increasing anthropogenic activities. Mostly, industrial sectors and other human activities like mining, incineration of coal and waste, and steel dispensation are major causes for increase in Zn concentration in the environment. Other sources of Zn contamination are crops or drinking water stored in metal chambers [23].
Nickel is the transition element that is present in environment only at very less amount and small doses are require for biological systems, but it can be hazardous when the concentration exceeds the permissible limit [23]. The higher concentration of Ni causes different types of cancer in various parts of animal body, mostly of those staying nearby industries [48]. It also causes damage to cells, reduces body weight, and damages the liver and heart [22]. Nickel is utilized in the steel industries, nickel-cadmium battery industries on a large scale, and in other metallic products [22, 48]. It is also found in paint formulation and cigarettes. The industrial wastes containing nickel enter into the water bodies contaminate the water and affect aquatic living organisms [49]. Earlier studies reported that microorganisms have problems for their growth and development in the presence of nickel. But, some studies in current days reported that some microorganisms have developed resistance to nickel [23].
Biofilms are communities of one or more species of microorganisms living within the protection of an extracellular matrix composed of polysaccharides, proteins, DNA, and other molecules, collectively termed as the extracellular polymeric substances (EPS) [12, 19, 22].
Microbial EPS is crucial for the formation of biofilm and cell aggregates, which contribute to protect cells from hostile environments and can bind significant amounts of heavy metals [53–56]. Biofilm and planktonic cells have distinct heavy metal and metalloid susceptibility [57–59]. It is suggested that the complexation or sequestration of heavy metals and retarding their diffusion in to the biofilm may be responsible for protecting cells from heavy metal toxicity [58]. Microbial EPS are also of particular interest and relevance to the bioremediation process due to their involvement in flocculation and binding of heavy metals from solutions [53, 60–62].
3.4 Interaction of Metal and Biofilm Bacteria
The availability of heavy metal ions is the hazardous factor for environment. Availability of metal ions in different components of environment like water, soil, microorganisms, aquatic lives, and other forms of lives are dependent on several factors like industrial activity, natural sources like volcanic eruption, and unlimited anthropogenic activities. Again, its presence, concentration and effects are influenced by several environmental factors and circumstances like pH, alkalinity, redox potential, and action of microorganisms. Earlier, it was thought that metals have toxic effect only on microbial metabolic process or mechanism. But later, it is discovered that they are not only lethal but their existence can persuade different mechanism of metal resistance in microorganisms. Microbes and metal ion communication can take place through different mechanisms. These mechanisms are classified depending on the pathway of communication of metal ions with microbes such as active and passive uptake of metal ions [63]. The biological interaction of microbes and metal ions transform the ions from toxic to less toxic or few accessible forms or arrest metal ions to inhibit their opening into bioprocess ( Figure 3.1). The various interactive mechanisms are available such as biosorption, bioleaching, biovolatilization, bioimmobilization, and bioaccumulation. The EPS of biofilm bacterial cell also interact with metal ions, due to communication among positive charge metal ions and negative charge EPS of cell surface [64].
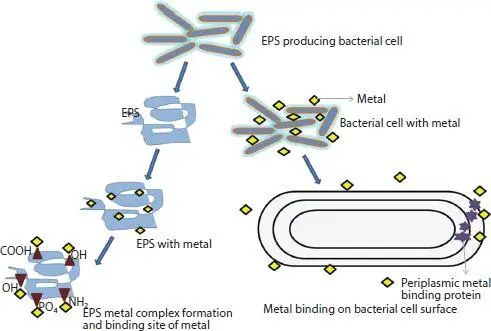
Figure 3.1 Interaction of metal with EPs and binding on bacterial cell surface.
3.5 Biodetoxification Mechanisms
The accumulation of heavy metals in food chain and their toxicity affects to biological system creates various problems. These can also enter to water bodies and contaminate soil through agricultural extract, industrial wastes, domestic runoff, and other commercial activities. We can eliminate or reduce heavy metal from contaminated sources. Therefore, there are different types of detoxification technology that have been utilized to eliminate heavy metals from contaminated sources. These detoxification technologies are briefly described as follows:
Читать дальше













