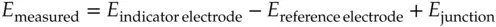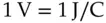In both voltammetry and potentiometry experiments, a difference in rates of diffusion associated with salt bridges used with reference electrodes leads to a higher flux for either positive or negative ions over those of the opposite charge. The excess of charge “pushes back” against continuing build‐up of charge leading to a steady state situation. The result is a net separation of charge and a junction potential or diffusion potential. Junction potentials are generally small, but they can be serious errors in potentiometric experiments. Later chapters discuss this issue in depth.
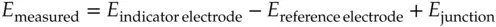
Electrical phenomena are associated with charged particles. Electrons are the most common charge carriers that one encounters, but ions in a solution are also important charge carriers. The purpose of this chapter is to define some electrochemical terms and introduce some fundamental concepts associated with electrical charge and phase boundaries.
All electrochemical techniques involve measuring (and sometimes manipulating) the voltage at an electrode. What is voltage? Voltage is a measure of the electrical energy available to do work on a charged particle. A charged particle has an electric field associated with it that interacts with its environment. An electric field is the force that two charged particles experience as a function of distance between them. Charges with the same sign repel each other and charges of opposite sign attract. Consequently, the arrangement of charged particles surrounding a given location will determine whether a charged particle coming into that place from the outside will be stabilized by net attractive forces or will be destabilized by net repulsive forces. The electric potential energy for a charged particle is defined as the energy spent or released in the process of inserting a positive test charge into a specific environment. For example, consider an arbitrary location in some material, such as point A shown in Figure 1.1.
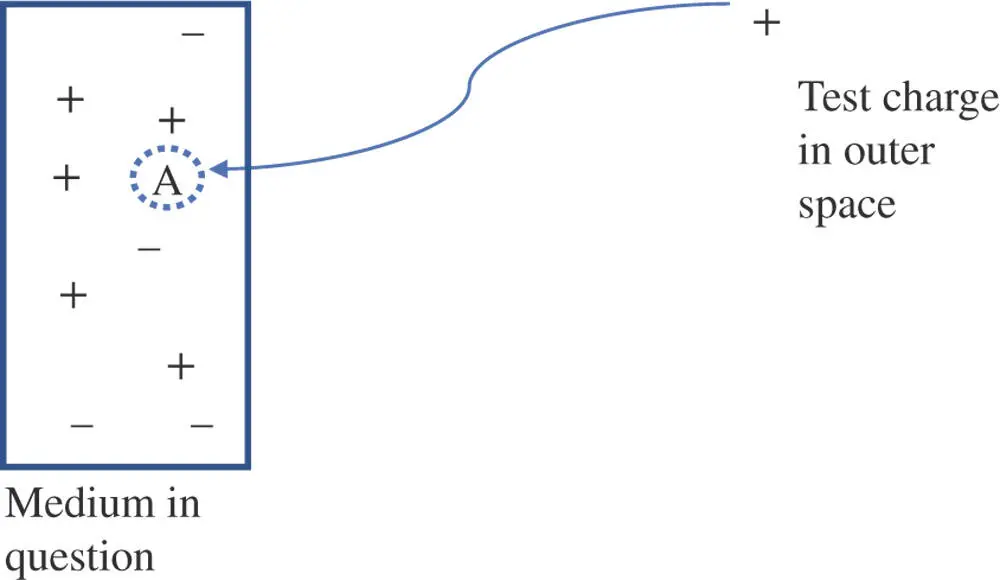
Figure 1.1The electric (or electrostatic) potential energy at a point, A, in a given medium is a measure of the net energy required or released in moving a test charge from outer space (where it is assumed to be free of forces to interact with) to point A where other charges attract or repel it.
There exists some collection of charges surrounding the point in question (point A in Figure 1.1). If one were to bring a positively charged particle from outer space, where it is assumed the test charge is free from the influence of any outside electromagnetic fields to point A, one would have to do work (energy would be spent) to overcome other positive charges in the neighborhood. However, if negative charges dominate the neighborhood at point A, there would be a net attractive force on the test charge and energy would be released in moving it from outer space to that position. The energy spent or released in moving a test charge from outer space to point A is the electric potential energy (also known as the electrostatic potential energy) at that point. For simplicity, this energy is often called the potential at point A. If a different arrangement of charges exists at point B (as in Figure 1.2), then moving a test charge from outer space to point B is associated with a different electric potential energy.
There is not a practical way of measuring the absolute electric potential energy at point A or at point B. However, it is possible to measure the electric potential energy difference between points A and B. A common strategy is to define some point in the system under study as a reference point. Then, the potential at any other point in the system is the electric potential energy difference between the point in question and the reference point. In this approach, no absolute electric potential energies need be evaluated. In the field of electronics, the reference point is often the electric potential energy of a conductor in direct contact with the earth (Figure 1.3). One might say that the electric potential of electrons in the ground is zero, but that is really just a statement about their relative energy; it does not represent an absolute value. (Furthermore, the ground is really just a local benchmark, because small variations in the electrical potential can be found at different places around the earth. Fortunately, a local reference is adequate for most practical situations.).
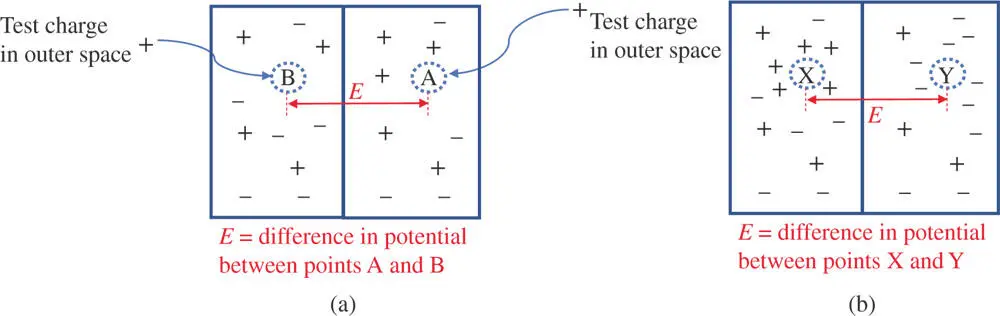
Figure 1.2a) The absolute electrical potential of a given point cannot be measured. However, the difference in potentials, E , between two points, A and B can be measured. In practice, some point within a system is defined as a point of reference so that the potential at any other point can be defined relative to reference point. b) The electric potential for a positive charge around X is much more positive than at Y driving a positive charge from X toward Y or driving a negative charge from Y to X.
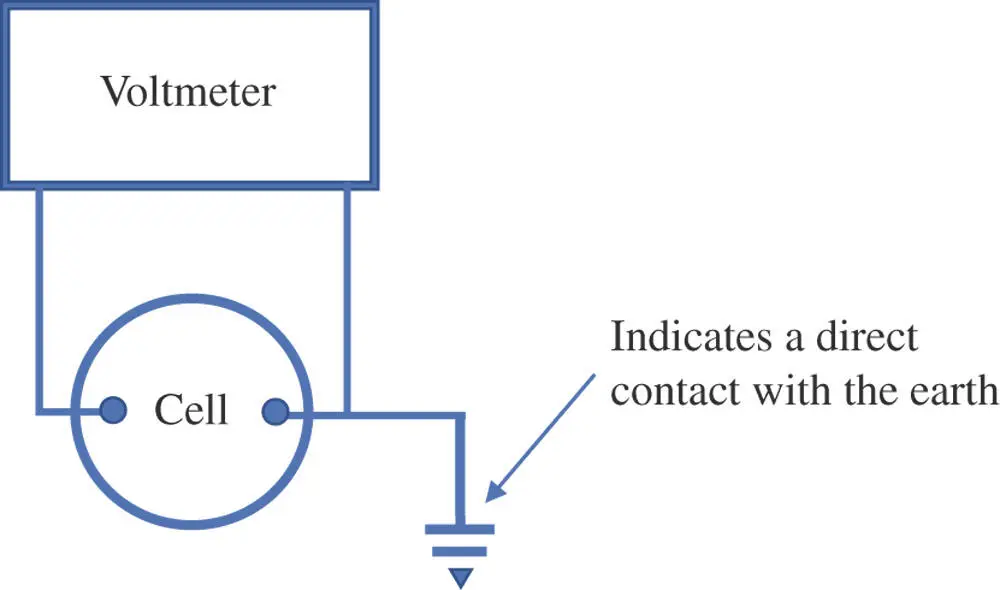
Figure 1.3Electronic circuits form a continuous loop including all components. Usually some point in the circuit is linked to a conductor that has direct contact with the earth. That point becomes a reference point and is treated as though its potential is 0 V.
In electrochemical experiments, the reference potential is established by the use of a reference electrode. It is common practice to refer to a potential or voltage at an electrode when in actuality the value being discussed is the electric potential difference between the electrode in question and the reference electrode being used in that experiment. In older literature, this potential is also called the electromotive force or EMF as it is the energy available to drive charges from one point to the other and do work. Properties of reference electrodes are discussed in chapter 2(section 2.3.4.3).
In the thought experiment described earlier, a single particle was used as a test charge. The standard definition for electric potential is the energy required to move a unit of positive charge to the position in question. The standard unit of charge in physics is the coulomb and the standard unit of energy is the joule. Electrical energy is measured in volts. One volt represents one joule per coulomb of charge moved.
(1.1) 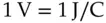
The volt is the unit of electric potential energy per unit charge that one normally uses with simple meters in the laboratory. So, whenever someone refers to a voltage at some part of their system, they are describing the electric potential difference between that point and some reference point (usually the ground). The voltage is the number of joules released or spent in moving a coulomb of charge from the reference point to the point in question.
It is important to remember that the potential is the electrical work done per unit charge. However, a coulomb is a rather large amount of charge compared to the charge on a single electron. If moving a coulomb of charge from point A to point B costs 1.00 J of energy, then how much energy is required to move a single positively charged particle between the same points? First, one can calculate charge in coulombs/electron using Faraday's constant, F , the number of coulombs per mole of electrons, 9.6485 x 10 4C/mol, together with Avogadro's number.
Читать дальше