Tarso B. Ledur Kist - Open and Toroidal Electrophoresis
Здесь есть возможность читать онлайн «Tarso B. Ledur Kist - Open and Toroidal Electrophoresis» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Open and Toroidal Electrophoresis
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:4 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
Open and Toroidal Electrophoresis: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Open and Toroidal Electrophoresis»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
The exact expressions of separation efficiency, resolution, peak capacity, and many other performance indicators of the open and toroidal layouts are presented and compared.
Featuring numerous illustrations throughout,
offers chapters covering: Solvents and Buffer Solutions; Fundamentals of Electrophoresis; Open Layout; and Toroidal Layout. Confronting Performance Indicators is next, followed by chapters on High Voltage Modules and Distributors; Heat Removal and Temperature Control; and Detectors. The book finishes with an examination of the applications of Toroidal Electrophoresis.
The first book to offer a detailed account of Toroidal Electrophoresis—written by one of its creators
Compares the toroidal layouts with the well-established open layouts of the three most used platforms (Capillary, Microchip, and Slab) Provides solutions to many of the experimental issues arising in electromigration techniques and discusses the voltage distributors and detectors that are compatible with the toroidal layouts Richly illustrated with a large number of useful equations showing the relationships between important operational parameters and the performance indicators
is aimed at method developers and separation scientists working in clinical analysis, and food analysis, as well as those in pharmacology, disease biomarker applications, and nucleic acid analysis using the Capillary, Microchip, or slab Platform. It will also benefit undergraduate and graduate students of inorganic analytical chemistry, organic analytical chemistry, bioanalysis, pharmaceutical sciences, clinical sciences, and food analysis.

 and has an important role in the Coulomb equation , equation 1.1. This constant is high for water and Table 1.1shows a list for comparison. Moreover, the average number of hydrogen bonds between water molecules in pure water is much larger than the average number of hydrogen bonds within hydrofluoridic acid or ammonia, due to the symmetry of the two sp 3bonding orbitals and the two sp 3non-bonding orbitals of water molecules. This maximizes the average number of intermolecular hydrogen bonds – up to two as negative charge donor and two as negative charge acceptor – totaling up to four hydrogen bonds per molecule. Therefore, pure water is liquid from 273.15 K up to 373.15 K at 1 atm. The electric force (
and has an important role in the Coulomb equation , equation 1.1. This constant is high for water and Table 1.1shows a list for comparison. Moreover, the average number of hydrogen bonds between water molecules in pure water is much larger than the average number of hydrogen bonds within hydrofluoridic acid or ammonia, due to the symmetry of the two sp 3bonding orbitals and the two sp 3non-bonding orbitals of water molecules. This maximizes the average number of intermolecular hydrogen bonds – up to two as negative charge donor and two as negative charge acceptor – totaling up to four hydrogen bonds per molecule. Therefore, pure water is liquid from 273.15 K up to 373.15 K at 1 atm. The electric force (  ) among ions within a liquid is given by:
) among ions within a liquid is given by:
 represents the electric permittivity of the vacuum,
represents the electric permittivity of the vacuum, 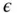 represents the permittivity of the liquid (solvent),
represents the permittivity of the liquid (solvent),  is defined as the relative permittivity,
is defined as the relative permittivity,  and
and  are the charges, and
are the charges, and  the distance from each other. Table 1.1shows the relative (static) permittivity of some solvents; note that water is among the highest.
the distance from each other. Table 1.1shows the relative (static) permittivity of some solvents; note that water is among the highest.
 C range and one atmosphere of pressure, except for water and hydrogen fluoride, which has a boiling point of 19.5
C range and one atmosphere of pressure, except for water and hydrogen fluoride, which has a boiling point of 19.5  C.
C. C. Extracted from Wohlfart and Lechner (2008) [3].
C. Extracted from Wohlfart and Lechner (2008) [3].
 C observed with the formamide derivatives at 20
C observed with the formamide derivatives at 20  C.
C.












