1 ...6 7 8 10 11 12 ...24 Some biological acids and their K avalues are listed in Table 1.1.1, together with a calculation of the proportion ionised at typical pH (7.4).
The calculation is done as follows (using acetic acid as an example):
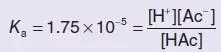
(where HAc represents undissociated acetic acid, Ac −represents the acetate ion). At pH 7.4, [H +] = 3 . 98 × 10 −8mol l −1 . Therefore,
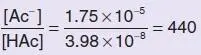
(i.e. the ratio of ionised to undissociated acid is 440:1; it is almost entirely ionised).
The percentage in the ionised form =  × 100% = 99 . 8% .
× 100% = 99 . 8% .
Table 1.1.1
| Acid |
Ka |
% ionised at pH 7.4 |
| Acetic, CH 3COOH |
1.75 × 10 −5 |
99.8 |
| Lactic, CH 3CHOHCOOH |
0.38 × 10 −4 |
99.9 |
| Palmitic acid, CH 3(CH 2) 14COOH |
1.58 × 10 −5 |
99.8 |
| Glycine, CH 2NH 2COOH (carboxyl group) |
3.98 × 10 −3 |
100 |
As stated earlier, polarity is not difficult to predict in organic molecules. It relies upon the fact that certain atoms always have electronegative (electron withdrawing) properties in comparison with hydrogen. The most important of these atoms biochemically are those of oxygen, phosphorus, and nitrogen. Therefore, certain functional groups based around these atoms have polar properties. These include the hydroxyl group (–OH), the amino group (–NH 2), and the orthophosphate group (–OPO 3 2−). Compounds containing these groups will have polar properties, whereas those containing just carbon and hydrogen will have much less polarity. The presence of an electronegative atom does not always give polarity to a molecule – if it is part of a chain and balanced by a similar atom this property may be lost. For instance, the ester link in a triacylglycerol molecule (discussed below) contains two oxygen atoms but has no polar properties.
Examples of relatively polar (and thus water- soluble) compounds, which will be frequent in this book, are sugars (with many –OH groups), organic acids such as lactic acid (with a COO −group), and most other small metabolites. Most amino acids also fall into this category (with their amino and carboxyl groups), although some fall into the amphipathic (‘mixed’) category discussed below.
Another important point about polarity in organic molecules is that within one molecule there may be both polar and non-polar regions. They are called amphipathic compounds. This category includes phospholipids and long-chain fatty acids ( Figure 1.4). Cell membranes are made up of a double layer of phospholipids, interspersed with specific proteins such as transporter molecules, ion channels and hormone receptors, and molecules of the sterol, cholesterol ( Figure 1.5). The phospholipid bilayer presents its polar faces – the polar ‘heads’ of the phospholipid molecules – to the aqueous external environment and to the aqueous internal environment; within the thickness of the membrane is a non-polar, hydrophobic region. The physicochemical nature of such a membrane means that, in general, molecules cannot diffuse freely across it: non-polar molecules would not cross the outer, polar face and polar molecules would not cross the inner, hydrophobic region. Means by which molecules move through membranes are discussed in Chapter 2(Box 2.1).
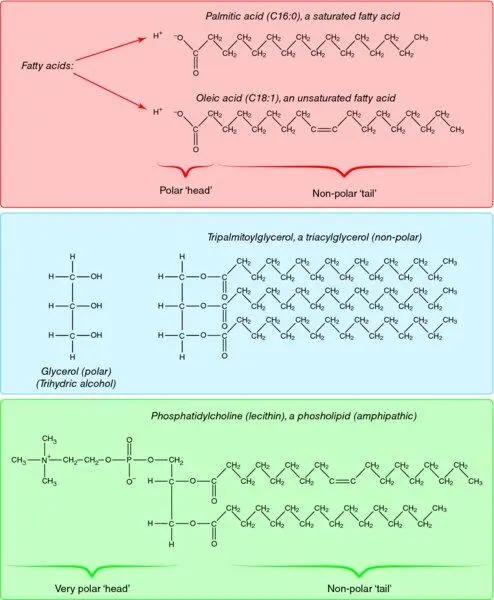
Figure 1.4 Chemical structures of some lipids. A typical saturated fatty acid (palmitic acid) is shown with its polar carboxylic group and non-polar hydrocarbon tail. Glycerol is a hydrophilic alcohol. However, it is a component of many lipids as its hydroxyl groups may form ester links with up to three fatty acids, as shown. The resultant triacylglycerol has almost no polar qualities. The phospholipids are derived from phosphatidic acid (diacylglycerol phosphate) with an additional polar group, usually a nitrogen-containing base such as choline (as shown) or a polyalcohol derivative such as phosphoinositol. Phospholipids commonly have long-chain unsaturated fatty acids on the 2-position; oleic acid (18:1 n -9) is shown.
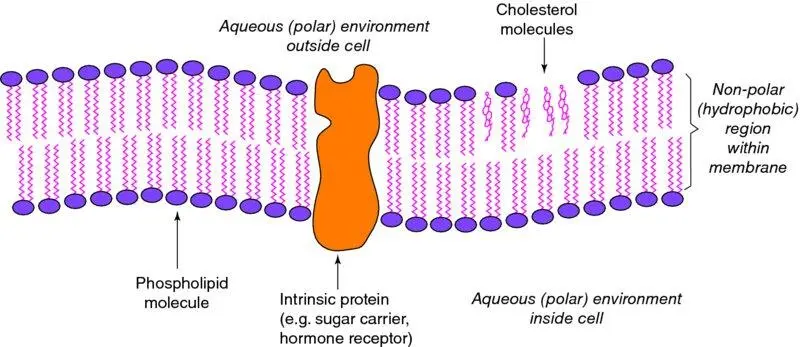
Figure 1.5 Structure of biological membranes in mammalian cells. Cell membranes and intracellular membranes such as the endoplasmic reticulum are composed of bilayers of phospholipid molecules with their polar head-groups facing the aqueous environment on either side and their non-polar ‘tails’ facing inwards, forming a hydrophobic centre to the membrane. The membrane also contains intrinsic proteins such as hormone receptors, ion channels, and sugar transporters, and molecules of cholesterol which reduce the ‘fluidity’ of the membrane. Modern views of cell membrane structure emphasise that there are domains, known as ‘rafts,’ in which functional proteins co-locate, enabling interactions between them. These lipid rafts are characterised by high concentrations of cholesterol and of certain phospholipids (glycosphingolipids).
The long-chain fatty acids fall into the amphipathic category – they have a long, non-polar hydrocarbon tail but a more polar carboxylic group head (–COO −). Another compound with mixed properties is cholesterol ( Figure 1.6); its ring system is very non-polar, but its hydroxyl group gives it some polar properties. However, the long-chain fatty acids and cholesterol may lose their polar aspects completely when they join in ester links. An ester is a compound formed by the condensation (elimination of a molecule of water) of an alcohol (–OH) and an acid (e.g. a carboxylic acid, –COO −). Cholesterol (through its –OH group) may become esterified to a long-chain fatty acid, forming a cholesteryl ester (e.g. cholesteryl oleate, Figure 1.6). The cholesteryl esters are extremely non-polar compounds. This fact will be important when we consider the metabolism of cholesterol in Chapter 10. The long-chain fatty acids may also become esterified with glycerol, forming triacylglycerols ( Figure 1.4). Again, the polar properties of both partners are lost, and a very non-polar molecule is formed. This fact underlies one of the most fundamental aspects of mammalian metabolism – the use of triacylglycerol as the major form for storage of excess energy.
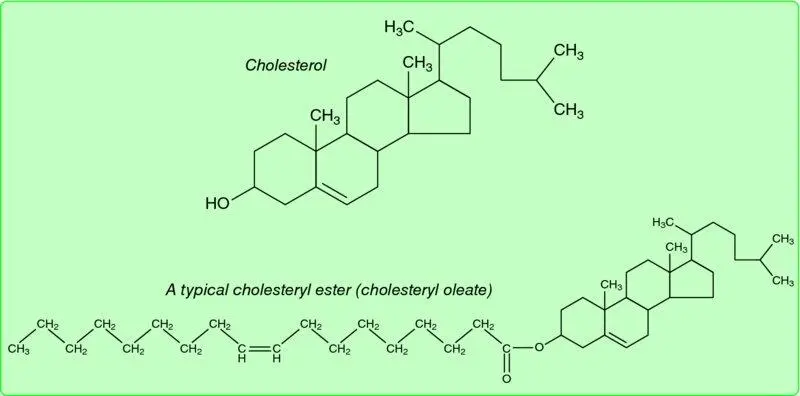
Figure 1.6 Cholesterol and a typical cholesteryl ester (cholesteryl oleate). In the structure of cholesterol, not all atoms are shown (for simplicity); each ‘corner’ represents a carbon atom, or else –CH or –CH 2. Cholesterol itself has amphipathic properties because of its hydroxyl group, but when esterified to a long-chain fatty acid the molecule is very non-polar.
Читать дальше

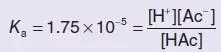
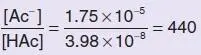
 × 100% = 99 . 8% .
× 100% = 99 . 8% .













