MOFs are synthesized by coordination of organic linker to inorganic units by strong bonds [12]. These connections between organic and inorganic building blocks through coordination interactions are such that they create vacant spaces (pores) between the each individual building block. As a result of such porosity, MOFs provide accessible pore volume in the bulk of the materials moreover than accessible area at the surface of material. Right selection of the building blocks makes it possible to vary some parameters, such as the pore size (to increase pore diameter to 98 Å), density (to decrease to 0.126 g·cm −3) and surface area (typically in range from 1,000 to 10,000 m 2·g −1) which are exceeding those of traditional porous materials such as zeolites and carbons [13]. In summary, porosity and surface area of MOFs could be tuned through right selection of organic ligands with correct size, flexibility and appropriate inorganic nodes.
The nature, strength and the number of coordination interactions between organic and inorganic building blocks of MOFs are the main reasons for evaluation of their stability. On one hand, selection of building blocks based on Hard-Soft acid-based theory is very beneficial for synthesis of highly stable MOFs [14]. For example hard metal ions such as Al(III) and Zr(IV) [15] could develop stable MOFs through connection with carboxylate-donor organic linker because these building blocks are hard Lewis acid and base, respectively. Another group of stable MOFs are based on selection of soft metal ions like late 3D metal ions and soft N-donor organic ligands like pyrazolate based linkers. On the other hand, the number of coordination bonds between inorganic nodes and organic linkers is another critical factor on the stability of MOFs. The higher number of coordination bonds, the higher stability of the MOF.
Another desirable character of MOFs is their crystalline structure. Selection of well-defined individual molecular building blocks could develop regular structure and periodic frameworks. As a result of their regular crystalline structure, we can tune their chemical and physical properties through logical designing of the framework and right selection of building blocks.
One of the most important advantages of MOFs from other conventional polymers or porous materials is the fact that we can tailor their chemical properties through rational choice of functional groups. Tunability in chemical functionality of MOFs could be provided via three functionalization strategies including selection of functional organic ligand, functionalization of inorganic nodes and functionalization of vacant spaces (pores) inside the framework of MOFs ( Figure 1.3) [14, 16]. These functions could be introduced prior to the self-assembly process by selection of desirable functional building blocks (pre-synthesis functionalization strategy) or after synthesis of the frameworks (post-synthesis functionalization strategy) [17, 18]. Unlimited ways for functionalization of MOFs is key factor to control their chemical properties and host–guest interactions. Definitely, we can state that tunability in chemical functionality and consequent ability in control over host–guest chemistry of MOFs has very essential roles in absorbing the attraction of scientist toward MOFs.

Figure 1.3 Three strategies for functionalization of MOFs.
Considering the diverse and useful characteristics of MOFs including regular crystalline structure, high porosity and surface area, hybrid inorganic–organic nature, satisfactory structural stability and tunability in chemical functionality, they are employed for different types of applications such as gas storage and separation [19–23], heterogeneous catalysis [24, 25] and photocatalysis [26–28], sensing [29–33], removal and separation of hazardous chemicals [34–36], drug delivery [37], bio sensing [38–41] and other medical applications [42] electrical conductivity [43] and electrochemical applications [44], ion storage and conductivity [45, 46], and designing MOF-based energetic materials [47]. Such diversity in chemical properties and application of MOFs show that intensive studies are necessary to identify the maximum capability of MOFs for each type of applications.
1.3 Functional Metal–Organic Frameworks
We mentioned that there are three different strategies for functionalization of MOFs. Among these methods, functionalization of organic ligand absorbed massive attention thanks to unlimited diversity in synthesis of organic ligands with different length and shape as well as rich host–guest chemistry of organic functional groups. So, functionalized organic linkers are extensively applied for construction of functional metal-organic frameworks (FMOFs) [14, 48].
The most important reason for introduction of organic functional groups into the framework of MOFs is to control the host–guest chemistry of FMOFs and gain selectivity to special guest molecules in presence of other analytes or tailoring the photoelectrochemical properties of FMOFs. However, moreover than domination in the host–guest chemistry and chemical properties of FMOFs, introduction of organic functional groups influence on the structural properties of FMOFs through induced structural changes and different types of secondary interactions ( Figure 1.4).
Although linker functionalization strategy gained lots of attention to control the chemical properties of FMOFs, FMOFs could be developed through other methods. For example a special kind of linker can be stabilized on inorganic nodes or special kind of functional materials like carbonitride, nanoparticles, dyes, graphene or graphene oxide or polyoxometalates can be incorporated into the pores of MOFs to fabricate a multi-functional MOF-based hybrid material ( Figure 1.5) [49]. In addition, synergic cooperation of functions in the structure of FMOFs is useful for improvement of practical application of FMOFs.
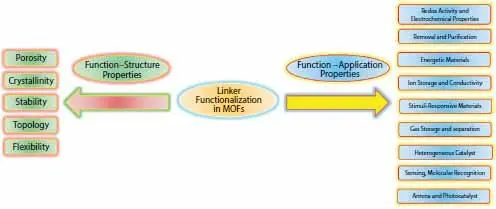
Figure 1.4 Function–structure and function–application properties of functional MOFs. Function–structure properties include relationship between structural features of MOFs and functional groups while function–application properties encompass the effects of functional groups on different type of applications.

Figure 1.5 Different application of functional or multi-functional MOF-based materials [16].
For a coherent and purposeful study, organic functional groups are sorted in two different methods. Based on first approach, organic functional groups are classified owing to their chemical properties and structural similarities. In this view, applied functional groups into the structure of FMOFs are categorized in four major groups including (I) nitrogenbased functions, (II) oxygen-based functions, (III) sulfur-based functions and carbonyl-based functions. All these four major groups entail a number of organic functional groups ( Figure 1.6). In addition to these four major groups of organic functional groups into the pores of FMOFs, there are some of other functions especially phosphonate and fluorine-based functions which are applied in the structure of FMOFs.
In the second approach, organic functional groups are classified based on their role in the structure of FMOFs. In this view functional groups are classified as coordinating sites ( Figure 1.7) and gust-interactive sites ( Figure 1.8). Functional groups as coordinating site are those which can coordinatively bond to metal ions during the synthesis of MOFs. Coordinating functional groups must be strong coordination bond donor with high chelation ability to develop stable FMOFs. There are two common types of coordinating functional groups. First common coordinating functions are O-donor functions such as sulfonate, phosphonate, enoxide and especially carboxylate. Other common coordinating functions are N-donor functions like pyridine, pyrazine and heterocyclic azole functions including pyrazole, triazole and tetrazole. Although, we mentioned that some functions like carboxylate, sulfonate, phosphonate, enoxide, pyridine, pyrazine and heterocyclic azole functions applied as coordinating sites, this does not mean that these functions did not apply as guest interactive sites. In other word, mentioned functions can apply as both coordinating and guest-interactive sites.
Читать дальше















