David Bowers - Understanding Clinical Papers
Здесь есть возможность читать онлайн «David Bowers - Understanding Clinical Papers» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Understanding Clinical Papers
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:3 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 60
- 1
- 2
- 3
- 4
- 5
Understanding Clinical Papers: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Understanding Clinical Papers»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Understanding Clinical Papers — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Understanding Clinical Papers», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:

Figure 2.3 Explaining the background to a research study.
Source: From Graham et al. (2000), © 2000, with permission from BMJ Publishing Group Ltd.
These questions will normally be discussed by reference to existing evidence . The Introduction to a paper is not the place to look for a comprehensive literature review, and introductory sections in most papers are brief, but there are one or two pointers to help you decide if the evidence is being presented in a fair and unbiased way:
Is there reference to a systematic review (see Chapters 10and 11)? Or if not, to a search strategy which the authors used to identify relevant evidence? For an example, see Figure 2.4, taken from a study of the association between birthweight and adult blood pressure.
Is the evidence mainly from the authors' own group or do the authors quote a range of evidence, even if it is not in support of their own views?

Figure 2.4 Introduction explaining the rationale for the study.
Source: Reproduced from: Shanahan et al. (2019).
Many clinical studies are carried out because the evidence is ambiguous or contradictory. Is there a dilemma which is posed by the evidence and is it clearly spelled out in the Introduction?
Generally speaking, the justification for a new study is that the existing evidence is unsatisfactory and a typical Introduction summarizes why, as in Figure 2.4from a study on the use of social media to post about self‐harm. The commonest justifications for new research are that:
Different studies have come to different conclusions about the topic and it isn't possible to come to an answer without new work.
The evidence cannot be applied in the setting being considered by the authors. For example, good evidence may cease to be of value simply because it is old – trials showing the benefit of treatment may no longer be useful if a disorder changes so that its sensitivity to treatment changes. Similarly, evidence from one part of the world cannot always be applied freely elsewhere.
The evidence may be incomplete. For example, we may know that rates of smoking are increasing among young women but we don't know why.
The evidence may be of poor quality.
If these elements of the Introduction are well presented, then it should be clear what the paper is about and why the authors have chosen to conduct the work that they have. Armed with this background briefing, you can now move on to check the specific objectives of the authors' work (see Chapter 3).
ETHICAL CONSIDERATIONS
Nearly all studies in healthcare that involve contact with people will require ethical approval. What that means is that the researchers will have had to submit their proposals to a panel of experts, such as a local research ethics committee, who decide whether the project is ethical or not. For example, the risks of any research should be outweighed by its benefits and participants should have been given the opportunity to participate or not as they wished, without their decision influencing their medical care.
Most authors will indicate that their study has been approved by the appropriate body governing research ethics – usually either in the Methods section or the Acknowledgements. Increasingly, authors will mention any particular ethical dilemmas raised by their research either in the Introduction or the Discussion of their paper. Where there are particular questions raised by a study, the authors may expand upon them (including, for example, details of the information given to participants and the way in which consent was obtained).
Certain types of research cause particular ethical concerns. For example, young children, or those with cognitive impairment or learning disability, or patients who are unconscious, cannot give consent to participate in research that nonetheless asks extremely important questions about clinical care. In these situations, researchers may undertake research with ethical approval, provided certain criteria are met (see Figure 2.5from a study of a weight management intervention in adults with a learning disability).

Figure 2.5 Ethical considerations in a study involving adults with a learning disability.
Source: Reproduced from Harris et al. (2017).
CHAPTER 3 The Aims and Objectives
Following the Introduction, you should look for a clear statement of the purposes of the current work. This statement can come in two forms: the aims of the study and the objectives.
Aims are general statements about purpose. For example, the authors might wish to examine the attitudes of hospital nurses to colleagues with mental health problems.
Objectives are specific questions, suggested by previous research or theory. For example, ‘Does taking the oral contraceptive pill increase the risk of stroke among women of childbearing age?' One particular sort of objective is to test an hypothesis.
Because the terminology of hypothesis testing is so widely used, we will start there.
HYPOTHESES
Often, studies will ask more than one question, so they will have several hypotheses. In these circumstances, you should look for a main hypothesis ( Figures 3.1and 3.2) and the other questions will form subsidiary or secondary hypotheses .

Figure 3.1 Statement of a study's main hypothesis.
Source: From Thompson et al. (2000), © 2000 Elsevier.
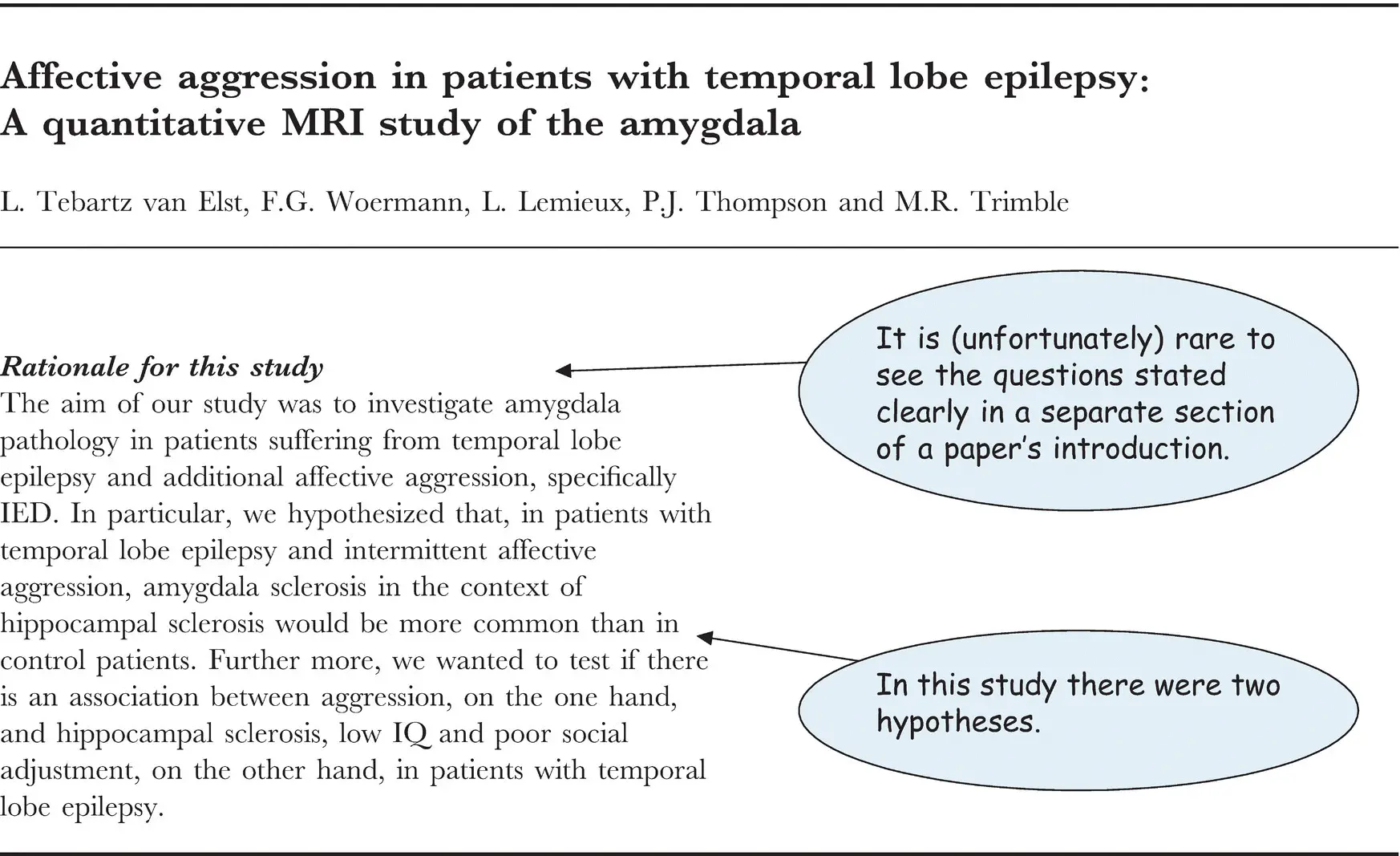
Figure 3.2 A study with two hypotheses.
Source: From Tebartz van Elst et al. (2000), © 2000 Oxford University Press.
There are important reasons why a study should have only one main question:
If a study tests many hypotheses, then just by chance it is likely to produce positive results for some of them. (See Chapter 31on hypothesis testing and the possibility of false‐positive results from multiple testing.)
We can trust a negative result only if we know that a study was large enough; otherwise, there is a possibility of false‐negative results. Many researchers therefore make an estimate of sample size to help them decide how big to make their study so that they can avoid this sort of error (see Chapter 13). To do that calculation they need to know what the main outcome of interest is, and the main outcome will be chosen to test the main hypothesis.
There used to be a conventional way of stating a study's hypothesis, which involved the use of a null hypothesis and the description of a study set up to disprove or refute an hypothesis . Although this approach is still sometimes taught, you will almost never come across examples in papers. The null hypothesis was a way of stating a question in the form ‘situation A is no different from situation B'. It arose because certain statistical tests operate by testing whether an assumption of similarity is likely to be true.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Understanding Clinical Papers»
Представляем Вашему вниманию похожие книги на «Understanding Clinical Papers» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Understanding Clinical Papers» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












