David Bowers - Understanding Clinical Papers
Здесь есть возможность читать онлайн «David Bowers - Understanding Clinical Papers» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Understanding Clinical Papers
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:3 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 60
- 1
- 2
- 3
- 4
- 5
Understanding Clinical Papers: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Understanding Clinical Papers»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Understanding Clinical Papers — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Understanding Clinical Papers», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
The inclusion of five new chapters containing this qualitative material is the most important change in our book from the second edition. We are very pleased to have been able to welcome an experienced qualitative researcher as a co‐author, who has contributed this new material.
In addition, we have, not surprisingly, taken the opportunity to update many of the examples of clinical papers with which we illustrate the ideas contained throughout the book. At the same time, we have sharpened and clarified the text were we felt it was needed. We have added small amounts of new material here and there – where we felt that these additions, drawn from our familiarity with the evolving health research literature, would improve the book.
The book should appeal, as before, to doctors, nurses, health visitors, physiotherapists, radiographers, dietitians, speech therapists, health educators and promoters, podiatrists, and all of those other allied professionals (and students in each of these disciplines) – and to all of those involved in health research.
David Bowers, Bridgette Bewick, Allan House, and David Owens
Leeds , 2013
Preface to the Fourth Edition
The third edition of this book was published in 2013: that's seven years ago! We are thankful that all of the previous editions were so well liked, and that we received so many favourable comments from readers. But seven years is a long time and we have felt for a while that the book needed refreshing to take into account readers’ suggestions and our own feelings about the potential for improvements. As a consequence, this fourth edition has been extensively revised and extended. The changes include four completely new chapters:
Studies of Complex Interventions.
Systematic Review of Qualitative Studies.
Research Using Already‐Collected Data. This new chapter will include material on missing data, confounding by indication, and other relevant topics.
Poisson Regression.
At the same time every other chapter has been extensively revised, and with much new material added, on, for example, the difference between clinical and statistical significance; reservations about the p‐value; the consequences of multiple testing and methods of correction; nested case–control studies; feasibility and pilot studies; cluster randomized trials; stratified random allocation; adaptive trial designs; sub‐group analysis; standardized guidelines for writing trials and reviews; and much more. In addition, we have improved the text throughout where we thought this would lead to better understanding. Importantly, we have also updated a great many of the examples from the literature.
As before, this edition of our book should appeal to all of those professionals involved in clinical work who wish to improve their understanding of the research literature.
David Bowers, Allan House, David Owens, and Bridgette Bewick
Leeds 2020
CHAPTER 1 Some Preliminaries
Before you start reading a paper, you could usefully ask one or two questions which help set the work in context:
Who wrote the paper?
In what sort of journal does the paper appear?
Who (and what) is acknowledged?
WHO WROTE THE PAPER?
Often, one person writes an article such as a review or an editorial. This is less common for papers describing the results of a research study. Because most research is a joint enterprise, papers describing research studies are usually published under the names of a number of people – the research team. From the list of authors, you can tell:
The range of expertise of the research team. Professional backgrounds of the authors (and sometimes their level of seniority) are often included, with the address of each.
The research centre or centres involved in the study. This is useful when you've been reading for a while and you know whose work to look out for – for whatever reason!
The principal researcher. He or she is often named first, or sometimes identifiable as the only author whose full address and contact details are listed (called the corresponding author).
Figure 1.1shows a typical example of a research project that required a collaborative effort.

Figure 1.1 Authors and research centres listed at the start of a research paper.
Source: Reprinted from Kuwawenaruwa et al. (2019) https://doi.org/10.1016/j.socscimed.2019.02.005
The list of authors may be quite long. The more people involved with a study, the less likely it is that one of them has a pre‐eminent position, so there may be no principal author. The authors may simply be listed in alphabetical order. They are listed at the top of research papers and (often) in guidance from the journal on how to cite the paper.
When a large study involving many sites is published, it may be that the work is written up by a small team, on behalf of the larger group. You may then find that there are some named authors, or only one or two, or (in very large studies) many authors are listed while the rest of the team is listed elsewhere – as in Figure 1.2. This type of multiple authorship is unavoidable if everybody is to get credit for participating in large studies, such as this project exploring the genetics of stroke.
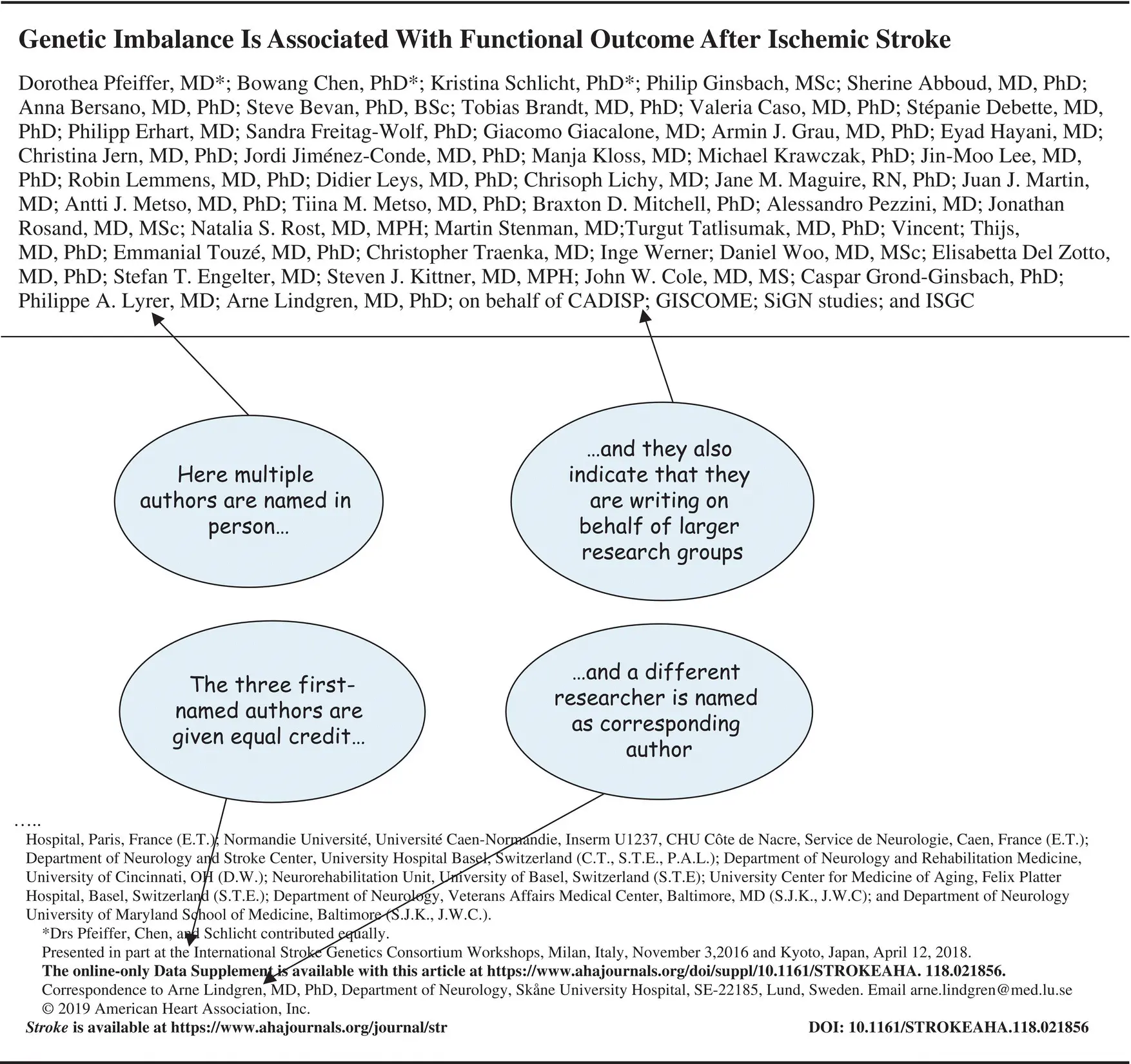
Figure 1.2 Paper written by multiple authors on behalf of several research groups.
Source: From Pfeiffer et al. (2019).
An undesirable form of multiple authorship arises if members of an academic department attach their names to a paper when they had nothing to do with the study. This is sometimes called ‘gift authorship', although it isn't always given very freely. To try to stop this practice, many journals now expect each author to explain exactly what part he or she has played in the study. For this, and other useful information, you should turn to the Acknowledgements at the end of the paper.
IN WHAT SORT OF JOURNAL DOES THE PAPER APPEAR?
Not all journals are the same. Some are mainly aimed at members of a particular professional group, and therefore include political news, commentaries, and personal opinions. Others publish only research articles which have not appeared elsewhere, while some aim to mix these functions.
In some journals, the letters pages are designed to allow readers to express their opinions about articles which have appeared in previous issues. In others, the letters pages contain only descriptions of original studies.
What appears in a journal is decided by the Editor, nearly always with the help and advice of an Editorial Committee. The best journals also seek opinions from external referees who comment on papers sent to them and advise on suitability for publication. Because these referees are usually experts in the same field as the authors of the paper, this process is called ‘peer reviewing'. It isn't always easy to tell whether papers for a journal are peer‐reviewed, which is unfortunate because the peer‐reviewing process is the best means of establishing the quality of a journal's contents. You shouldn't trust the results of any data‐containing study if it appears in a journal which does not use the peer‐reviewing system.
Some journals produce supplements , which are published in addition to the usual regular issues of the main journal. They may be whole issues given over to a single theme or to describing presentations from a conference or symposium. Often they are produced (unlike the main journals) with the help of sponsorship from pharmaceutical companies. Papers in these supplements may not have been reviewed by the same process as papers in main journals and for that reason they tend not to be of as high quality.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Understanding Clinical Papers»
Представляем Вашему вниманию похожие книги на «Understanding Clinical Papers» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Understanding Clinical Papers» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












