Table 5.9 Platelet additive solution component concentration (mM).
Sources : Modified from Gulliksson H. Platelet additive solutions: current status. Immunohematology 2007; 23(1):14–17; Alhumaidan H, Sweeney J. Current status of additive solutions for platelets. J Clin Apher 2012; 27:93–98; Vermeij J. Methods for preparing platelet products. WO 2016/014854 A1. Geneva: World Intellectual Property Organization International Bureau, January 28, 2016.
|
InterSol a(Fenwal) |
Isoplate a(Terumo) |
Composol(Fresenius) |
Plasma Lyte A (Baxter) |
SSP+(MacoPharma) |
| Sodium chloride |
77 |
92.7 |
90 |
90 |
69 |
| Potassium chloride |
0 |
5 |
5 |
5 |
5 |
| Magnesium chloride |
0 |
1.5 |
1.5 |
3 |
1.5 |
| Sodium citrate |
10 |
0 |
11 |
0 |
10 |
| Sodium phosphate buffer |
26 |
0.5 |
0 |
0 |
26 |
| Sodium acetate |
30 |
27.2 |
27 |
27 |
30 |
| Sodium gluconate |
0 |
22.9 |
23 |
23 |
0 |
aCurrently the only platelet additive solutions approved for use in the United States.
5.4 Platelet concentrates—whole blood
Description of component
The official term for this component is platelets. These are platelets suspended in plasma or platelet additive solution (PAS) ( Table 5.9) prepared by centrifugation of WB. Platelets may also be produced by cytapheresis (see Chapter 12). A unit of WB‐derived platelets must contain at least 5.5 × 10 10platelets [24]. Although there is no required volume, these units usually have a volume of about 50 mL to maintain viability and function during storage.
To prepare a dose of pooled platelets, 4–6 units of WB‐derived platelets are pooled together in an open system. After platelet pooling in open system, the platelets have a short outdate of 4 hours at room temperature. In addition, these platelet products are tested for bacterial contamination prior to issuing with point of care testing [65]. These platelets can be leukoreduced and irradiated if it is required based on the patient’s diagnosis. Acrodose platelet is an alternative technology for pooling platelets in the closed system [66]. Acrodose platelets are pretested for bacterial contamination, do not require pooling in the hospital blood bank, and can be issued in emergency situations immediately without further manipulation. Acrodose platelets can be stored for a total of 5 days at room temperature.
WB‐derived and apheresis platelets can be suspended in PASs. Advantages of PASs include reduction in allergic transfusion reactions, possibly TRALI and ABO‐mismatched hemolysis [67–69]. PAS also is used to mitigate deleterious effects of pathogen‐inactivated technology and can potentially improve viability of platelets and extend their days of storage. There are multiple PAS products on the European and US markets for WB‐derived and apheresis platelets.
There are two methods for preparing platelets from WB: the platelet‐rich plasma (PRP) method and the buffy coat method ( Figure 5.3) [70]. In the United States, platelets are prepared using the PRP method; in Europe and Canada, the buffy coat method is used [70, 71]. The PRP method uses low g‐forces (“soft” spin), and the PRP easily separates from the red cells [72] within 8 hours after blood collection. The PRP is transferred into a satellite bag to separate it from the red cells ( Figures 5.2and 5.3). The PRP is then centrifuged at higher g‐forces (“hard” spin) and the platelet‐poor plasma is removed, leaving a platelet concentrate and about 50 mL of plasma ( Figure 5.3). After the plasma is removed, the platelet concentrate is left undisturbed or, preferably, placed on the platelet storage rotating device for 1 hour to minimize platelet damage and allow for spontaneous resuspension [73]. In the PRP method, the first step is a soft spin and the second step is a hard spin ( Figure 5.3); because of the soft spin, about 20% of the plasma and 20–30% of the platelets remain with the red cells [70]. Another 5–10% of platelets are lost during the second centrifugation step when the PRP is converted to a platelet concentrate. Thus, the PRP method yields about 60–75% of the original platelets, a red cell unit containing about 40 mL of plasma, and about 50% or more of the leukocytes in the original unit of WB. The disadvantages of this method are the loss of some plasma that could be used for fractionation and the high leukocyte content of the platelets.
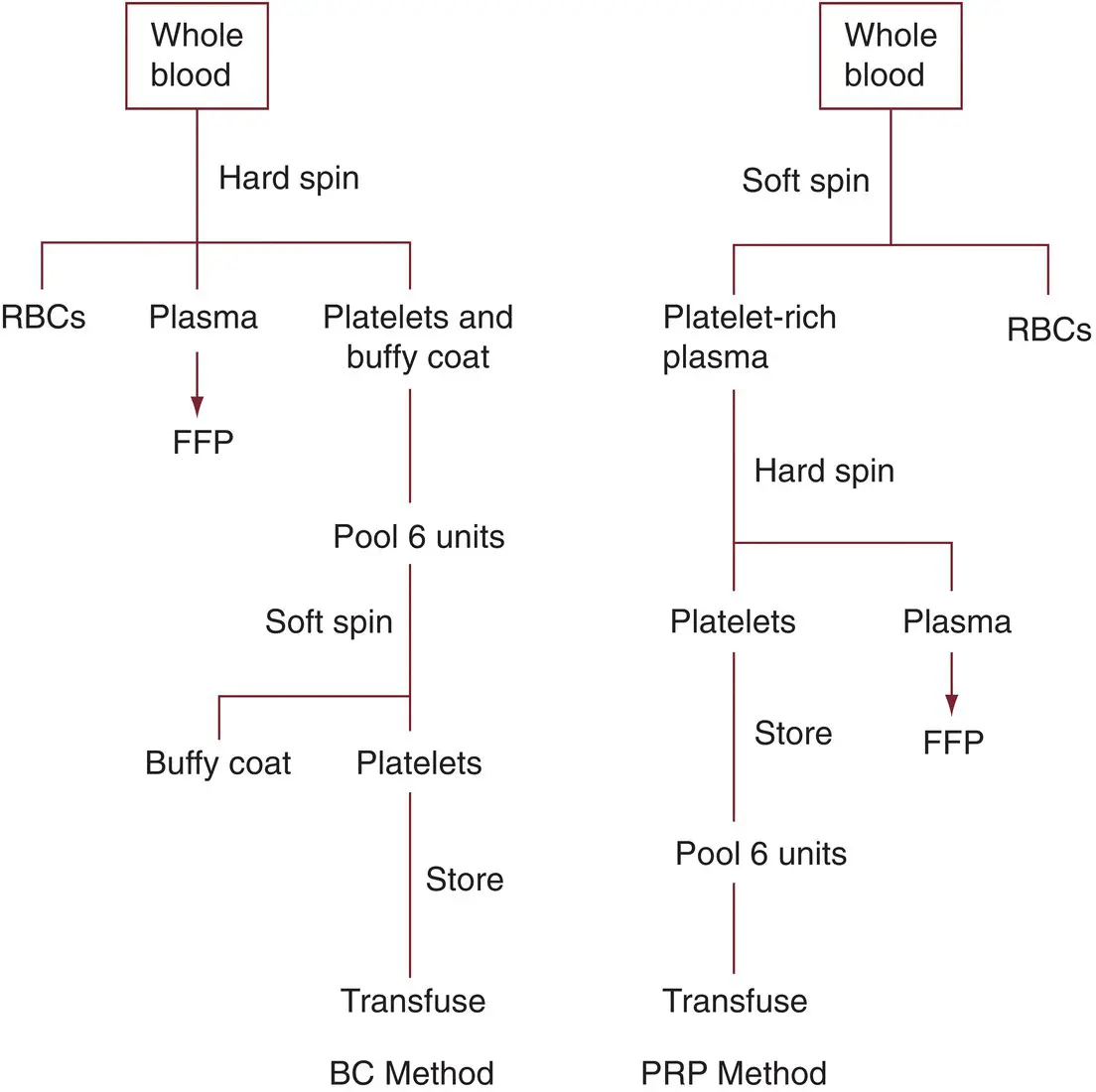
Figure 5.3 Comparison of platelet‐rich plasma (PRP) and buffy coat (BC) methods of platelet preparation. RBC, red blood cell.
When platelets are produced by the buffy coat method, the WB is centrifuged at a higher g‐force (hard spin) to create a buffy coat that also contains most of the platelets (85%) and leukocytes [70, 74]. Because the WB centrifugation step involves higher g‐forces, the red cells are more tightly packed and more plasma is obtained along with the buffy coat. However, to obtain most of the buffy coat, it is necessary to remove some of the red cells, and so there is a loss of about 20–25 mL of red cells [70]. To obtain a platelet concentrate, the buffy coat is centrifuged using low g‐forces, and the platelets are separated from the leukocytes and red cells. Thus, in this method, the first step is a hard spin and the second step a soft spin—the opposite of the PRP method. It is thought that the use of the soft spin in the second centrifugation may result in platelets that function better than those obtained by the PRP method, in which the second centrifugation is a hard spin when there is less WB to “cushion” the platelets [74, 75]. The effectiveness of the second centrifugation step is improved if several units of buffy coat are pooled, usually in groups of six [74]. When units of buffy coat are pooled for the second centrifugation, they may be suspended in an artificial platelet preservation (platelet additive) solution that improves the separation and the quality of platelets during storage [76]. Also during the second centrifugation step, the platelets are passed through a filter as they are separated, thus removing most of the leukocytes and producing a leukocyte‐depleted platelet component.
Storage conditions and duration
Platelets prepared by either method can be stored in either plasma or PAS. Platelets stored at 20–24°C maintain functional effectiveness for several days [13, 77–80]. Many variables affect the quality of platelets during storage. In addition to temperature, these other variables include the anticoagulant–preservative solution, storage container, type of agitation, anticoagulant, and volume of plasma [72, 81–83]. Gentle horizontal agitation is preferable to end‐over‐end agitation [83]. If continuous agitation is interrupted, platelets stored for up to 5 days maintain appropriate in vitro characteristics for up to 24 hours of interruption of agitation [84]. The composition, surface area, and size of the storage container influence the ability for carbon dioxide to diffuse out and oxygen to enter the platelet concentrate, and storage containers specifically designed to optimize platelet quality are now used routinely [85, 86].
Maintenance of the pH greater than 6.0 is the crucial factor indicating satisfactory platelet preservation. This combination of storage container, agitation, preservative solution, temperature, and the use of about 50 mL of plasma provides satisfactory preservation of platelets for up to 7 days [85, 87]. However, several instances of bacterial contamination of platelet concentrates stored for this period were reported [87, 88], and the storage time was reduced to the 5 days currently used [24]. The problem of bacterial contamination still exists (see Chapter 16), although recently with additional point of care testing for bacterial detection, the FDA approved storage of apheresis platelets up to 7 days [89] (see Chapter 7). The storage container needs to be FDA approved for apheresis platelet storage up to 7 days. In addition, large‐volume delayed sampling and secondary cultures can be used to extend platelet storage to 7 days [89].
Читать дальше