These trials are often termed Phase III trials in contrast with Phase I and Phase II trials which are concerned with early stages of the (often pharmaceutical) development process. Although the trials differ in aspects of their design, the majority have the general structure of a two (or more) group parallel design in which eligible patients are assigned to receive the alternative options (often treatments but more generally termed interventions) and then at some later time assessed in a way which will be indicative of (successful) outcome. The outcomes measured in these trials include the following: survival time, gastric emptying time, reduction in disease activity, visual field status, recurrent parasitaemia, major adverse cardiac events, pain, the number of hip fractures, systolic blood pressure and standard criteria used to assess dental restorations. In the trial of homoeopathic arnica for pain relief following hand surgery, assessment was made in a double‐blind or double‐masked manner in which neither the patient nor the assessor was aware of the specific treatment option actually received.
The methods used for the allocation to the options included simple randomisation of equal numbers per group, a 2 to 1 allocation; a minimisation procedure taking into account patient characteristics, randomisation to nursing homes (clusters) rather than to individual residents. For the split‐mouth design used for the comparing dental implants the authors’ state:
For randomization of the implant type, a pregenerated random sequence was created … . Opaque envelopes were sealed according to pregenerated list. An independent judge prepared all envelopes. … an assistant indicated which implant had to be placed first following the indications contained in the sequentially number envelope.
The non‐random allocation to a single‐arm study using a new bioabsorbable stent for coronary scaffolding might now be regarded as a feasibility study although the trial results were compared to that from historical data.
The trials ranged in size from 20 patients with colonic cancer to 5102 women with HER2‐positive breast cancer. One trial involved 522 eyes from 271 subjects another 88 single implant‐supported crowns teeth in 34 partially edentate patients. Although not fully detailed in the above summaries, methods of statistical analysis ranged from a simple comparison of two proportions to relatively complex methods using techniques for survival time outcomes.
In general, trials are designed to establish a difference between the (therapeutic) options under test and were one to exist. Consequently, they are sometimes termed superiority trials. However, in certain circumstances, as in the trial for the treatment of uncomplicated falciparum malaria, the research team were looking for non‐inferiority implying that the two treatment strategies of AQ + SP and AL would give very similar risks of failure. In the event, the trial suggested that AL was (unacceptably) less effective than AQ + SP implying that non‐inferiority was not established. Such designs usually imply that a satisfactory outcome is that the test treatment does not perform worse than the standard to an extent predefined by the investigating team. Thus, use of a non‐inferiority design often implies that, although some therapeutic loss may be conceded on the main outcome variable, other factors favouring the new therapy will have some features ( gain ) to offset this. For example, if the new compound was a little less effective (not equal to) but had a better toxicity profile, then this might be sufficient to prefer it for clinical practice.
1.3 Choice of design
1.3.1 Biological variability
Measurements made on human subjects rarely give exactly the same results from one occasion to the next. Even in adults, our height varies a little during the course of the day. If one measures the blood sugar levels of an individual on one particular day and then again the following day, under exactly the same conditions, greater variation in this than that of height would be expected. Hence were such an individual to be assessed and then receive an intervention (perhaps to lower blood sugar levels) any lowering recorded at the next assessment cannot necessarily be ascribed to the intervention itself. The levels of inherent variability may be very high so that, perhaps in the circumstances where a subject has an illness, the oscillations in these may disguise, at least in the early stages of treatment, the beneficial effect of the treatment given to improve the condition.
Example 1.13 Patient‐to‐patient variability – atopic eczema
The considerable between patient variability in the trial of Example 1.7is illustrated in Figure 1.1. In the 41 patients receiving Azathioprine, the reduction in disease activity (SASSAD) ranged from −10 to 32. There is considerable overlap of these values with those from the 20 patients receiving Placebo whose values range from −12 to 20. This figure clearly illustrates that, although there is considerable variation, the majority of patients in both groups improve. Further, the corresponding reduction in percentage body area affected with Azathioprine was reported to range from approximately −15 to 85% and for placebo approximately −20 to 45%. Nevertheless, even with the majority of patients improving in both groups, the trial of Meggitt, Gray and Reynolds (2006) indicated a better outcome, on average, for those receiving Azathioprine.
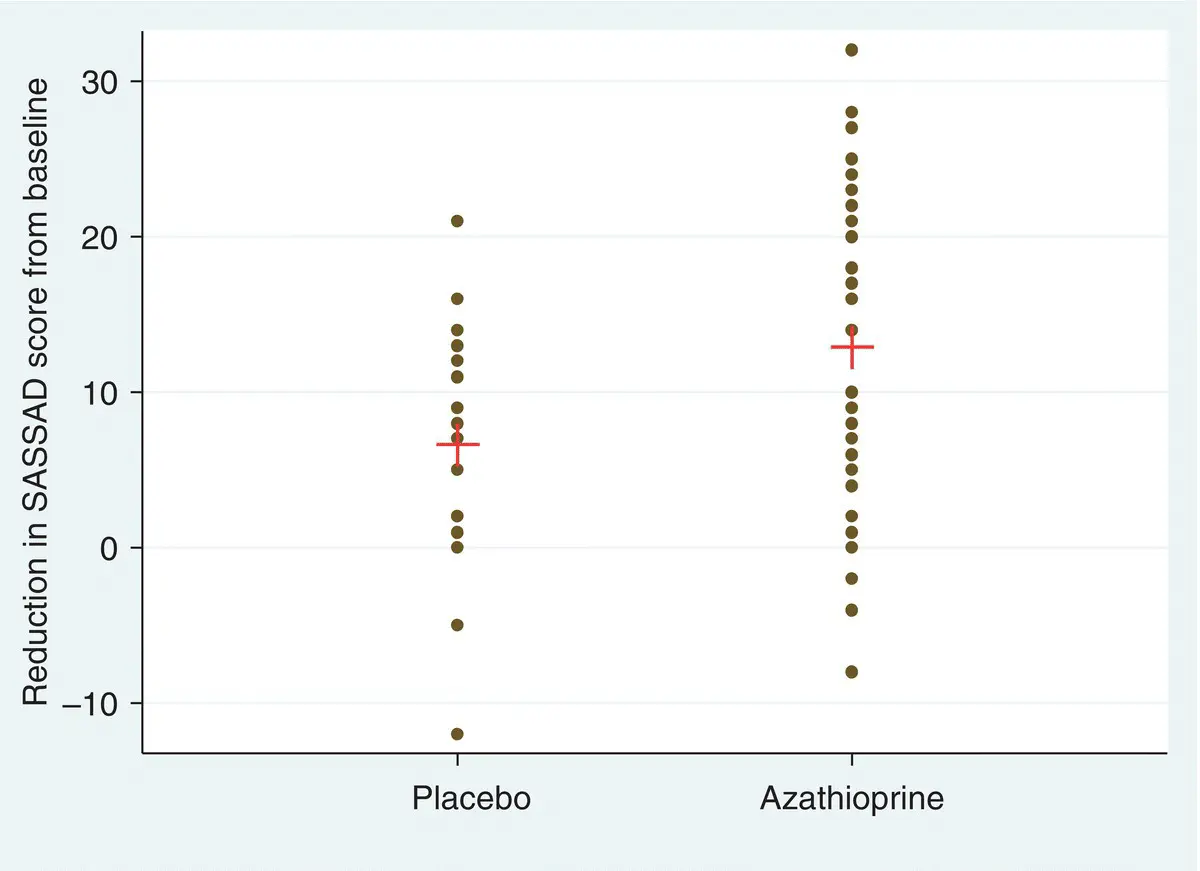
Figure 1.1Individual patient reductions in disease activity (SASSAD) for the Azathioprine and Placebo treatment groups with the corresponding means indicated.
Source: Data from Meggitt, Gray and Reynolds (2006).
With such variability, it follows that, in any comparison made in a biomedical context, differences between subjects or groups of subjects frequently occur. These differences may be due to real effects, random variation or both. It is the job of the experimenter to decide how this variation should be taken note of in the design of the ensuing trial. The purpose being that, once at the analysis stage, the variation can be partitioned suitably into that due to any real effect of the interventions on the difference between groups and that from the random or chance component.
Ronald A Fisher (1890–1962) in laying the foundations of good experimental design, although in an agricultural and biological context, advocated the use of randomisation in allocating experimental treatments. Thus, for example, in agricultural trials various plots in a field are randomly assigned to the different experimental interventions. The argument for randomisation is that it will prevent systematic differences between the allocated plots receiving the different interventions, whether or not these can be identified by the investigator concerned, before the experimental treatment is applied. Then, once the experimental treatments are applied and the outcome observed, the randomisation enables any differences between treatments to be estimated objectively and without bias. In these and many other contexts, randomisation has long been a keystone to good experimental design.
The need for random allocation extends to all experimental situations including those concerned with patients as opposed to agricultural plots of land. The difficulty arises because clinical trials (more emotive than experiments) do indeed concern human beings who cannot be regarded as experimental units and so should not be allocated the interventions without their consent. The consent process clearly complicates the allocation process and, at least in the past, has been used as a reason to resist the idea of randomisation of patients to treatment. Unfortunately, the other options, perhaps a comparison of patients receiving a ‘new’ treatment with those from the past receiving the ‘old’, are flawed in the sense that any observed differences (or lack thereof) may not reflect the true situation. Thus, in the context of controlled clinical trials, Pocock (1983) concluded, many years ago and some 30 years after the first randomised trials were conducted, that:
Читать дальше