Magma Redox Geochemistry
Здесь есть возможность читать онлайн «Magma Redox Geochemistry» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Magma Redox Geochemistry
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 100
- 1
- 2
- 3
- 4
- 5
Magma Redox Geochemistry: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Magma Redox Geochemistry»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Magma Redox Geochemistry
Volume highlights include: Magma Redox Geochemistry
The American Geophysical Union promotes discovery in Earth and space science for the benefit of humanity. Its publications disseminate scientific knowledge and provide resources for researchers, students, and professionals.
Magma Redox Geochemistry — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Magma Redox Geochemistry», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
List of Illustrations
1 Chapter 1 Figure 1.1 E‐pH diagram reporting the stability of water at T = 25°C and P =... Figure 1.2 The E‐pH (Pourbaix) diagram for the Fe‐S‐H 2O system at 25°C at 1 ... Figure 1.3 Limit of equilibrium potential‐pO 2–graphs in molten alkali... Figure 1.4 Log f O 2‐ Ph diagrams for 290°C (left panel) and 145°C (right pan... Figure 1.5 (a)Ellingham diagram for the main components of the melt/slag (s... Figure 1.6 Common solid oxygen buffers used in petrology and geochemistry. T... Figure 1.7 Two‐redox potential f O 2‐ f S 2diagram. The conformation of stabilit...
2 Chapter 2 Figure 2.1 Range of oxygen fugacity estimates for Solar Nebula, achondrites,... Figure 2.2 Log f O 2(normalized to the FMQ buffer) calculated for mantle perid... Figure 2.3 (a) V/Sc and (B) calculated Log f O 2(normalized to the FMQ buffer)...
3 Chapter 3 Figure 3.1 Locations of samples compiled in this study as a function of tect... Figure 3.2 Distribution of f O 2recorded by volcanics globally in different t... Figure 3.3 Distribution of f O 2recorded by mantle lithologies (peridotites a... Figure 3.4 V/Yb ratios of ridge (gray “+” symbols, Gale et al., 2013), back‐... Figure 3.5 Progressive degassing of a C–O–H–S vapor in ridge, arc, and plume... Figure 3.6 Magmatic oxygen fugacity as recorded by volcanics for samples in ... Figure A1 Temperatures and values of f O 2from the model of Ghiorso and Evans... Figure A2 XANES spectra of MORB glass VG3385 with spectra of mid‐ocean ridge... Figure A3 The measured experimental furnace f O 2in log units relative to the...
4 Chapter 4 Figure 4.1 Positions of redox buffers at 1 bar. From Frost, (1991) unless ot... Figure 4.2 Conceptual model of subduction zone cycling. Figure 4.3 Location of volcanic arcs and spreading ridges mentioned in the t...
5 Chapter 5Figure 5.1 (a) Solidus curves for peridotite in dry conditions (black line) ...Figure 5.2 Two depictions of the reduced and oxidized fluid realms for COH f...Figure 5.3 Illustration of the f O 2levels at which HRM and CRM occur, relate...Figure 5.4 Illustration of pressure effects on the extent of redox fractiona...Figure 5.5 Ranges of redox states found in various geodynamic settings and i...Figure 5.6 Illustration of redox fronts that develop between lithosphere and...Figure 5.7 Comparison of likely widely applicable peridotite solidi in Phane...
6 Chapter 6Figure 6.1 (a) Oxidized/reduced iron (log‐form) for simple binaries at 1400°...Figure 6.2 (a) Comparison of Paul and Douglas (1965) data with those of Tang...Figure 6.3 (a) S VI/S totfrom empirical formulations available in the literat...Figure 6.4 Mutual interaction of iron and sulfur redox couples in dry and hy...Figure 6.5 Patterns of sulfur release and their effect on melt sulfur distri...Figure 6.6 Covariation of sulfate and sulfide contents in andesitic to rhyol...Figure 6.7 (a) Covariation of sulfate and sulfide fractions in melt inclusio...Figure 6.8 (a) Plot of logK 15vs log f O 2, showing the nearly negligible effec...Figure 6.9 (a) Melt sulfate/sulfide ratios vs. dissolved water contents from...
7 Chapter 7Figure 7.1 Histograms of measurements of the Fe 2+/Fe Tratio in global survey...Figure 7.2 A simple model of olivine‐liquid equilibria and FeO‐MgO systemati...Figure 7.3 PRIMELT3 outputs from calculation of primary magma parental to th...Figure 7.4 PRIMELT3 prediction for the Mg# of the liquidus olivine that firs...Figure 7.5 PRIMELT3 predictions for the Mg# of the liquidus olivine that fir...Figure 7.6 Predictions of the pMELTS model of oceanic crustal production for...Figure 7.7 rhyoliteMELTS calculations showing the fraction of original liqui...
8 Chapter 8Figure 8.1 Histogram of the log(abundance) of 78 elements (M; bin size 0.5) ...Figure 8.2 log(Fe 3+/Fe 2+) as a function of f O 2, relative to the quartz‐fayal...Figure 8.3 Fe 3+/∑Fe as a function of f O 2, relative to the QFM buffer, predic...Figure 8.4 (a) log Fe 2O 3(in wt%) of MORB glasses determined by Berry et al....
9 Chapter 9Figure 9.1 XANES spectra from Cr 2+and Cr 3+standard glasses. There are seve...Figure 9.2 Adapted from Bell et al. (2017); illustrates how the crystallogra...Figure 9.3 Cr 2+/∑Cr ratios plotted vs. experimental f O 2from Roedder and Rey...Figure 9.4 Contains plots the XANES measured the Cr 2+/∑Cr ratios from quench...Figure 9.5 Cr partitioning data compiled from several studies plotted vs. ex...Figure 9.6 XANES measured Cr 2+/∑Cr values of a set of experimentally grown o...
10 Chapter 10Figure 10.1 Comparison between values of 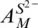 from O’Neill and Mavrogenes (2002...Figure 10.2 The relationship between S 6+/∑S and Fe 3+/∑Fe, estimated by combi...Figure 10.3 (a) Observed values of the sulfide capacity,
from O’Neill and Mavrogenes (2002...Figure 10.2 The relationship between S 6+/∑S and Fe 3+/∑Fe, estimated by combi...Figure 10.3 (a) Observed values of the sulfide capacity, 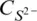 , at 1400°C of sil...Figure 10.4 Comparison between measured values of the activity coefficient o...Figure 10.5 The oxygen contents as the component FeO in sulfide mattes coexi...Figure 10.6 The effect of pressure on the SCSS, comparing Equation 10.43 of ...Figure 10.7 SCSS for a binary compositional join between FeO‐free “matrix” a...Figure 10.8 The effect of H 2O on SCSS. Data from Moune et al. (2009) at 1045...Figure 10.9 Sulfur saturation in ocean floor basaltic (OFB) glasses from Jen...Figure 10.10 The difference between [S] obsand the calculated SCSS from the ...Figure 10.11 Calculated atomic Ni/Fe (a) and Cu/Fe (b) in sulfide liquid in ...Figure 10.12 Comparison with the SCSS model of Smythe et al. (2017). (a) Cal...Figure 10.13 (a) Reproducibility of S analyses in basaltic glasses: a compar...Figure 10.14 Comparison of model [S] SCSSwith [S] obsfor Macquarie Island ba...Figure 10.15 Comparison between model [S] SCSSand [S] obsfor two sets of gla...Figure 10.16 Olivine‐hosted melt inclusions from Le Voyer et al. (2017). (a)...Figure 10.17 Ni and Cu abundances in ocean floor basaltic (OFB) glasses vers...Figure 10.18 Calculated [S] SCSSvs. [S] obsfor olivine‐hosted melt inclusion...Figure 10.19 (a) log 10 f S 2and (b) log 10 f SO 2calculated for 35 sulfide‐satu...
, at 1400°C of sil...Figure 10.4 Comparison between measured values of the activity coefficient o...Figure 10.5 The oxygen contents as the component FeO in sulfide mattes coexi...Figure 10.6 The effect of pressure on the SCSS, comparing Equation 10.43 of ...Figure 10.7 SCSS for a binary compositional join between FeO‐free “matrix” a...Figure 10.8 The effect of H 2O on SCSS. Data from Moune et al. (2009) at 1045...Figure 10.9 Sulfur saturation in ocean floor basaltic (OFB) glasses from Jen...Figure 10.10 The difference between [S] obsand the calculated SCSS from the ...Figure 10.11 Calculated atomic Ni/Fe (a) and Cu/Fe (b) in sulfide liquid in ...Figure 10.12 Comparison with the SCSS model of Smythe et al. (2017). (a) Cal...Figure 10.13 (a) Reproducibility of S analyses in basaltic glasses: a compar...Figure 10.14 Comparison of model [S] SCSSwith [S] obsfor Macquarie Island ba...Figure 10.15 Comparison between model [S] SCSSand [S] obsfor two sets of gla...Figure 10.16 Olivine‐hosted melt inclusions from Le Voyer et al. (2017). (a)...Figure 10.17 Ni and Cu abundances in ocean floor basaltic (OFB) glasses vers...Figure 10.18 Calculated [S] SCSSvs. [S] obsfor olivine‐hosted melt inclusion...Figure 10.19 (a) log 10 f S 2and (b) log 10 f SO 2calculated for 35 sulfide‐satu...
11 Chapter 11Figure 11.1 Isobaric calculations of H 2O and CO 2contents in Basaltic melt a...Figure 11.2 Variation of the log f O 2values as a function of the temperature...Figure 11.3 Experimentally determined S solubility data of basaltic melts as...Figure 11.4 Sulfur valence state at f O 2, expressed relative to FMQ. Figure m...Figure 11.5 Influence of oxygen fugacity on the partitioning of sulfur betwe...
12 Chapter 12Figure 12.1 Examples of iron concentration ([FeO] equivalent) in various ign...Figure 12.2 Variation of the Fe 3+/Fe TOT(Fe TOT= Fe 2++ Fe 3+) redox ratio as...Figure 12.3 Iron oxidation state in K 2O‐Al 2O 3‐SiO 2‐FeO melts at 1673 K in ai...Figure 12.4 Examples of compositional effects on the oxidation state of iron...Figure 12.5 Viscosity of (a) anorthite‐diopside (An‐Di) eutectic melt as a f...Figure 12.6 Viscosity of SiO 2, NaAlSi 3O 8and NaFeSi 3O 8melts.Figure 12.7 Isothermal viscosity as a function of the oxidation state of iro...Figure 12.8 Effects of the crystal (a) and water (b) contents on the viscosi...Figure 12.9 Viscosity, crystallinity, and oxidation state of andesite and ba...
13 Chapter 13Figure 13.1 Mössbauer spectra of an Fe‐bearing diopside glass prepared at 15...Figure 13.2 Example of Optical Absorption spectra of glasses containing diff...Figure 13.3 Optical absorption spectrum, in the range 4000–28500 cm –1,...Figure 13.4 Optical Absorption spectrum of a V‐doped sodium aluminosilicate ...Figure 13.5 Energy scaled K‐edge XAS spectrum of vanadium in divanadium trio...Figure 13.6 XANES spectra of first‐ row multivalent transition metal oxides ...Figure 13.7 K‐edge XANES spectra of Mn (a) and Fe (b) crystalline compounds ...Figure 13.8 Cerium L III‐edge XANES spectra for two Ce crystalline materials ...Figure 13.9 Schematic drawing of a lava, and XAS spectra at the Fe K‐edge an...Figure 13.10 Raman spectrum of an iron‐free pyroxene glass (CaO‐SiO 2‐Na 2O‐Mg...Figure 13.11 Raman spectra at room temperature for a series of iron‐pyroxene...Figure 13.12 a,b: Deconvolution of the 0.09 and 0.99 Raman spectra (based on ...Figure 13.13 Comparison of iron ratios analyzed by different techniques, wet...Figure 13.14 (a) Evolution of the normalized XANES spectra at the Fe K‐edge a...Figure 13.15 Fe 3+/Fe Totredox as different temperatures, 412, 618, 710 °C fr...
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Magma Redox Geochemistry»
Представляем Вашему вниманию похожие книги на «Magma Redox Geochemistry» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Magma Redox Geochemistry» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












