Figure 1.5. Molecular structure of 5CB (pentyl cyanobiphenyl).
In general, optical properties of liquid crystals fall into two distinct categories: those characteristics of individual constituent molecules and those unique to the bulk crystalline mesophases. Here we discuss optical properties that are largely decided by the individual constituent molecules; optical properties associated with various ordered mesophases of liquid crystals are elaborated in later chapters.
1.2.1. Electronic Optical Transitions and UV Absorption
Since liquid crystal constituent molecules are quite large, their energy level structures are rather complex. In essence, the basic quantum mechanical theory is similar to the one described in Chapter 10for a multilevel molecule. Generally, the energy levels are referred to as orbitals: π , n , and σ orbitals for the ground and low‐lying levels and π *, n *, and σ *for their excited counterparts. Since most liquid crystals are aromatic compounds, containing one or more aromatic rings, the energy levels or orbitals of aromatic rings play a major role. In particular, the π → π *transitions in a benzene molecule have been extensively studied. Figure 1.6shows three possible π → π *transitions in a benzene molecule.
In general, these transitions correspond to the absorption of light in the near‐UV spectral region (≤200 nm) [2]. These results for a benzene molecule can also be used for interpreting the absorption of liquid crystals containing phenyl rings. On the other hand, in a saturated cyclohexane ring or band, usually only σ electrons are involved. The σ → σ *transitions correspond to absorption of light of shorter wavelength (≤180 nm) in comparison to the π → π *transition mentioned previously.
These optical properties are also related to the presence or absence of conjugation (i.e. alternations of single and double bonds, as in the case of a benzene ring). In such conjugated molecules, the π electron’s wave function is delocalized along the conjugation length, resulting in absorption of light in a longer wavelength region compared to, for example, that associated with the σ electron in compounds that do not possess conjugation. Absorption data and spectral dependence for a variety of molecular constituents, including phenyl rings, biphenyls, terphenyls, tolanes, and diphenyl‐diacetylenes, may be found in [2].
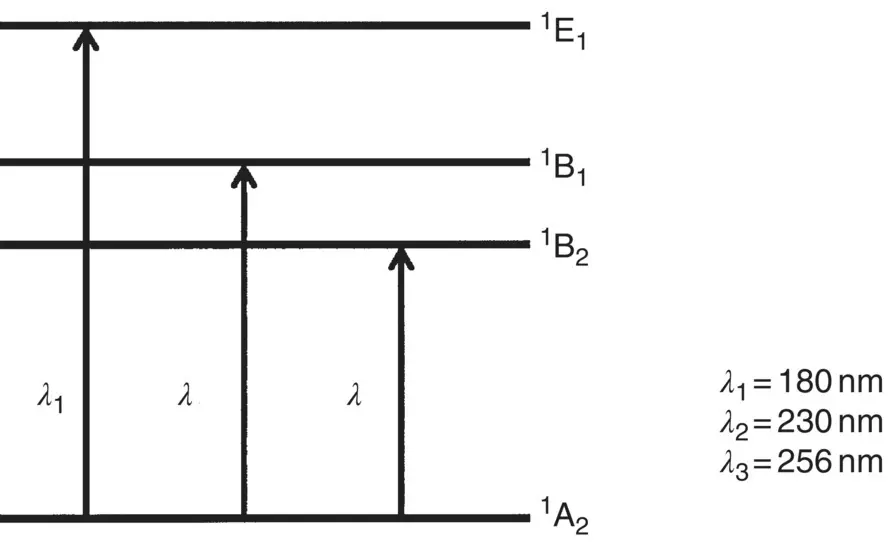
Figure 1.6. The π → π *electronic transitions in a benzene molecule.
1.2.2. Visible and Infrared Absorption; Terahertz, Microwave
The spectral transmission dependence of two typical liquid crystals is shown in Figure 1.7a and b. Liquid crystals are quite absorptive in the UV region, as are most organic molecules/liquids. In the visible and near‐infrared (IR) regime (i.e. from 0.4 to ~2 μm), there are no absorption bands, and thus liquid crystals are quite transparent in this regime. In the mid‐IR (3–5 μm) and IR (9–12 μm), rovibrational transitions begin to dominate, and liquid crystals are quite absorptive in this spectral area ( Table 1.1).
Table 1.1 Anisotropic Physical Parameters of a Typical Nematic Liquid Crystals (E7)
| n // |
n ⊥ |
Δ n |
α //(cm −1) |
α ⊥(cm −1) |
|
| 1.75 |
1.525 |
0225 |
… |
… |
( λ = 0.589 μm) |
| 1.71 |
1.50 |
0.21 |
… |
… |
( λ = 1.55 μm) |
| 1.70 |
1.49 |
0.21 |
55 |
40 |
( λ = 10.59 μm) |
| 1.73 |
1.57 |
|
0.9 |
3.1 |
(0.2 THz) |
| 1.76 |
1.62 |
|
7 |
27 |
(2 THz) |
Thermal diffusion constant:  |
Elastic constants:  |
Viscosity coefficients: (at 20 °C)  |

Figure 1.7. Transmission spectra of nematic liquid crystals: (a) 5CB; (b) N ‐(4‐methoxybenzylidene)‐4‐butylaniline (MBBA).
The absorption coefficient α in the UV (~0.2 μm) regime is on the order of 10 3cm −1; in the visible (~0.5 μm) regime, α ≪10 0cm −1; in the near‐IR (~3–5 μm and ~9–12 μm) regime, α ~ 10 2cm −1. There are, of course, large variations among the thousands of liquid crystals that have been synthesized. It is possible to identify liquid crystals with the desirable absorption/transparency for a wavelength of interest. For example, the terahertz and microwave regime, [5, 6] have identified/synthesized liquid crystals (LC’s) with relatively low absorption coefficient α ~ 10 0cm −1; low absorption is important since applications in such long‐wavelength regimes require much thicker interaction length (cell thickness).
A unique advantage of liquid crystals is the sizeable birefringence throughout the entire visible–IR‐terahertz‐microwave spectrum. At the 20–60 GHz region, [6] has shown that for a typical liquid crystal such as E7, ε e= 3.17 (refractive index n e= 1.78) and ε 0= 2.72 (refractive index n 0= 1.65), i.e. a birefringence Δ n ~ 0.13.
1.3. LYOTROPIC, POLYMERIC, AND THERMOTROPIC LIQUID CRYSTALS
There are several distinct types of liquid crystals: lyotropic, polymeric, thermotropic, and discotic. These materials exhibit liquid crystalline properties as a function of different physical parameters and environments such as temperature, molecular constituents’ structure, and concentration.
1.3.1. Lyotropic Liquid Crystals
Lyotropic liquid crystals are obtained when an appropriate concentration of material is dissolved in some solvent. The most common systems are those formed by water and amphiphilic molecules (molecules that possess a hydrophilic part that interacts strongly with water and a hydrophobic part that is water insoluble) such as soaps, detergents, and lipids. Here the most important variable controlling the existence of the liquid crystalline phase is the amount of solvent (or concentration). There are quite a number of phases observed in such water‐amphiphilic systems, as the composition and temperature are varied; some appear as spherical micelles, and others possess ordered structures with 1‐, 2‐, or 3‐D positional order.
Читать дальше


















