In addition to overcoming the elastic properties of the lungs and the chest wall, during active breathing the muscles of respiration also have to overcome the frictional forces opposing flow up and down the airways.
Site of maximal resistance
It is generally understood that resistance to flow in a tube increases sharply as luminal radius (r) decreases (with laminar flow, resistance is inversely proportional to r 4). It seems rather contradictory, therefore, to learn that in a healthy individual, the greater part of total airway resistance is situated in the large airways (larynx, trachea and main bronchi) rather than in the small airways. This is in part due to the fact that the flow velocity is greatest and flow most turbulent in the central airways, but also due to the much greater total cross‐sectional area in the later generations of airway ( Fig. 1.6). Remember, we only have one trachea, but by the 10th division we have very many small airways, which effectively function in parallel.
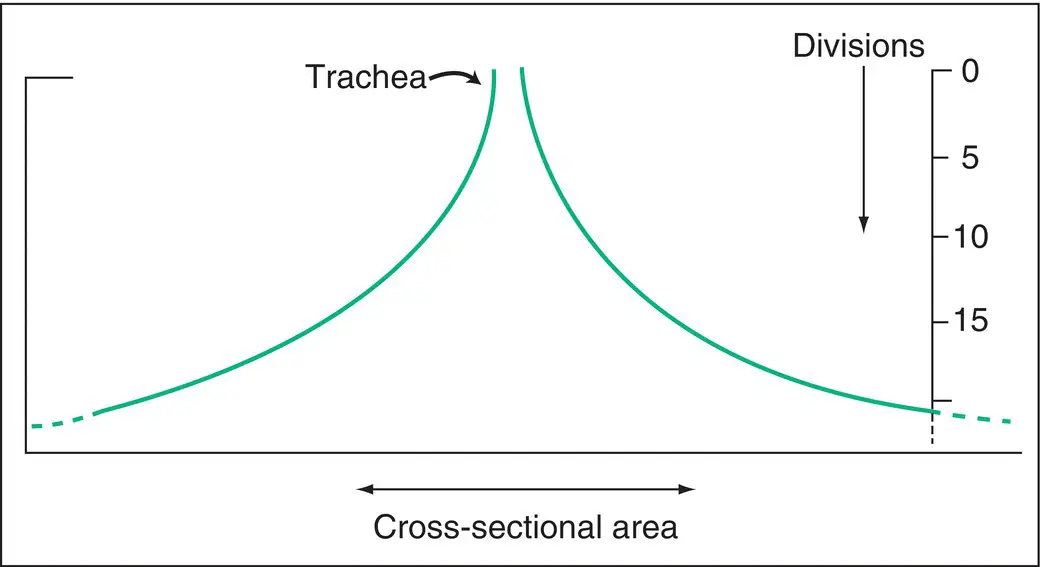
Figure 1.6 Diagrammatic representation of the increase in total cross‐sectional area of the airways at successive divisions.
Conditions may be different in disease states. Asthma and COPD – diseases that affect airway calibre – tend to have a greater proportionate effect on smaller generations of airway. The reduced calibre of the smaller airways then becomes overwhelmingly important and the site of principal resistance moves distally.
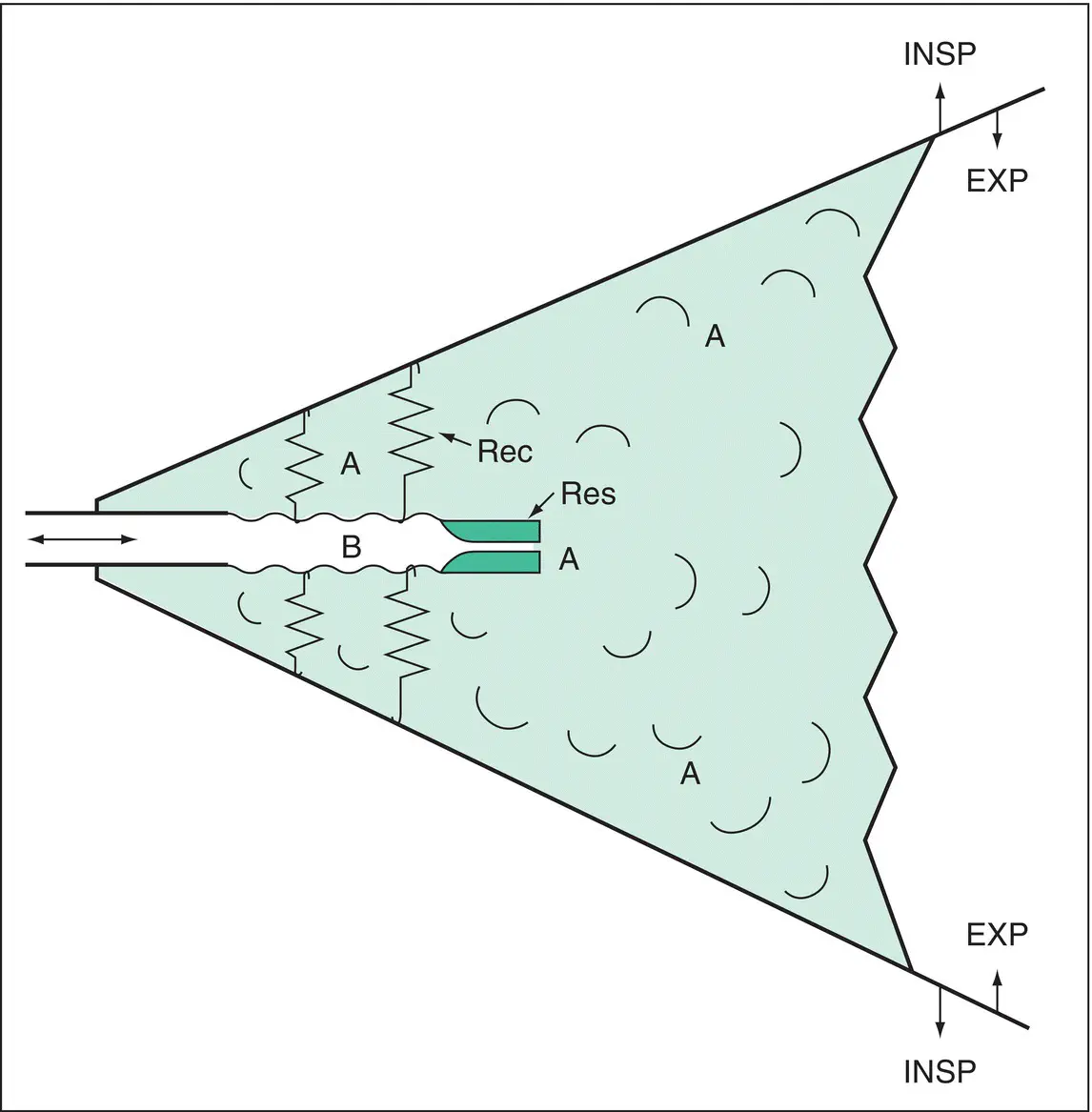
Figure 1.7 Model of the lung, demonstrating the flow‐limiting mechanism (see text). The chest is represented as a bellows. The airways of the lungs are represented collectively as having a distal resistive segment (Res) and a more proximal collapsible or ‘floppy’ segment. The walls of the floppy segment are kept apart by the retractile force of lung recoil (Rec). EXP, expiration; INSP inspiration.
Consider the model of the lung represented in Fig. 1.7. Here, the tube represents a route through generations of airways from the alveoli to the mouth. The smaller generations, without cartilaginous support, are represented by the ‘floppy’ segment (B). Airways are embedded within the lung and are attached externally to lung tissue whose elastic recoil and ultimate connection to the chest wall supports the floppy segments. This recoil force is represented by the springs.
During expiration, a positive pressure is generated in the alveolar space (A). Air flows from A along the airway, past B, where the pressure is lower (it must be, otherwise the air would not have flowed in this direction), and on to the mouth, where the pressure is nominally ‘zero’.
The pressure difference across the walls of the floppy segment (A minus B) would tend to cause this part of the airway to collapse. It is prevented from doing so by the retractile force of lung recoil (tension within the springs).
The flow‐limiting mechanism
During expiration, the extent of the pressure drop between A and B is proportional to the flow rate. Clearly, with increased effort, the pressure at A will increase, the pressure difference between A & B will increase and flow rate will be increased … up to a point. Eventually, a critical flow rate will be reached, where the pressure gradient between A and B is sufficient to overcome the retractile force of the lung, the airway wall collapses and airflow ceases. Once there is no flow, the pressure inside the airway at point B quickly equilibrates with that at A. With no pressure difference forcing the airway wall to collapse, the retractile force of the lung reopens the airway and flow recommences. This brings us back to where we started and the cycle begins again. It will be apparent that this mechanism determines a maximum possible flow rate along the airway. Any attempt to increase flow rate (associated with a greater pressure difference A to B) will simply result in airway closure. As each route out of the lung will similarly have a maximal possible flow rate, the expiratory flow from the lung as a whole will have an absolute limit. It can be seen that this limit (to expiratory flow) is set by the internal mechanics of the lung, not by muscular strength or effort (above a certain level of effort). That is perhaps fortunate; if it were not the case then lung function tests such as peak expiratory flow rate(PEFR) would not be tests of lung function at all, but of muscular strength.
The effects of disease on maximum flow rate
In asthma (see Chapter 10), airway narrowing occurs, leading to a greater resistance between point A (the alveolus) and point B. The pressure drop, A to B, for any given flow rate will therefore be greater than in the healthy lung, and the critical (maximal) flow rate (when the pressure difference between A and B is just enough to overcome the retractile force of the lung) will be lower. You may have known for some time that peak expiratory flow is reduced in asthma, but now you understand why.
In COPD (see Chapter 11), the loss of alveolar walls (emphysema) reduces the elastic recoil of the lung. There is therefore less protective retractile force on the airway wall and the critical pressure drop along the airway required to cause airway collapse will occur at a lower flow rate. Thus, maximum expiratory flow is also reduced in COPD.
Airway resistance and lung volume
It can easily be seen in the model that, as lung volume decreases, lung elastic recoil (tension within the springs) diminishes, providing less and less support for the floppy airway. It is clear, therefore, that the maximum flow rate achievable is dependent on lung volume and is reduced as lung volume is reduced. For any given lung volume, there will be a maximum expiratory flow that cannot be exceeded, no matter what the effort. You can confirm this by inspecting the shape of a flow loop, which is effectively a graph of the maximal flow rate achievable at each lung volume (see Chapter 3). A true PEFR can only be achieved by beginning forced expiration from a position of full inspiration. I would suggest you’ve been aware of this fact for longer than you realise. Immediately prior to blowing out the candles on your second birthday cake, you probably took a big breath in. At the age of 2, you had an intuitive understanding of the volume dependence of maximal expiratory flow rate.
Lung volume and site of maximal airway resistance
As we have already discussed, the greater part of airway resistance resides in the central airways. These airways are well supported by cartilage and so generally maintain their calibre even at low lung volumes. The calibre of the small airways, without cartilaginous support, is heavily dependent on lung volume. At lower lung volumes, their calibre is reduced, and resistance is increased. During expiration, therefore, as lung volume declines, the site of principal resistance moves from the large central airways to the small peripheral airways. The PEFR (see Chapter 3) tests expiratory flow at high lung volume and is therefore determined largely by the central airways. The forced expiratory volume in 1 second(FEV 1; see Chapter 3) is also heavily influenced by the central airway, though not as much as PEFR. Specialised lung function tests that measure expiratory flow at lower lung volumes (e.g. FEF 25‐75and 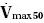 ; see Chapter 3) therefore provide more information about the smaller airways.
; see Chapter 3) therefore provide more information about the smaller airways.
Читать дальше



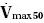 ; see Chapter 3) therefore provide more information about the smaller airways.
; see Chapter 3) therefore provide more information about the smaller airways.









