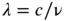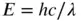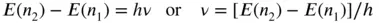At the center of a black hole is thought to be a singularity or singularity ring where density is infinite. This can be regarded as some of the most extreme matter in the Universe.
These extreme types of matter are not very relevant for life, although as we have seen, that has not stopped speculation about possible life forms inhabiting objects made of these types of matter. These ideas aside, one reason for investigating these unusual types of matter is that they remind us that the ordinary matter from which known life is made is a tiny subset of the matter in the known Universe. If ordinary matter constitutes say 5% of the matter in the Universe and 99% of that is plasma, then the gases, liquids, and solids that make up biological systems are made from about 0.05% of the types of matter in the Universe.
3.14 The Interaction Between Matter and Light
Ordinary matter has many complex interactions with radiation belonging to different regions of the electromagnetic spectrum. Here we look at one important interaction: the absorption and emission of electromagnetic radiation, which is of special importance to astrobiologists. The absorption and emission of light tell us about the structure of other stars, galaxies, and the interstellar medium. The absorption of light is used to identify gases in the atmospheres of exoplanets and will eventually be used to search for life. We discuss these approaches to the study of exoplanets and the search for life in more detail in Chapter 20. These wide uses of matter–light interactions mean that it is essential for us to grasp some of the basic concepts. In this last section of this chapter, we investigate the crucial insights needed for astrobiology. Later in this book, these concepts appear again as we investigate diverse challenges such as studying the habitability of other planets and the search for complex carbon compounds in the interstellar medium.
Light can be understood as a wave with a wavelength, λ (Figure 3.24). The frequency with which the peaks or troughs of those waves pass a given point is the frequency of light, ν , and the relationship between these two properties is given by:
(3.1) 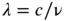
where c is the speed of light.
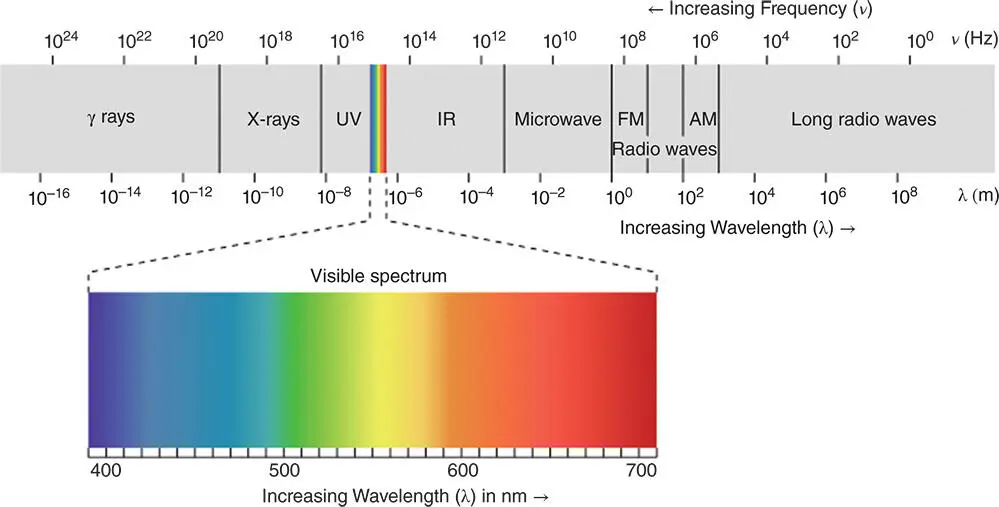
Figure 3.24 The wavelength and frequency of different types of electromagnetic radiation.
Source: Reproduced with permission of wikicommons.
The energy of light, in joules, at any given wavelength is given by:
(3.2) 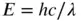
where λ is the wavelength of the light, and h is Planck's constant (6.626 × 10 −34J s). We can re-write this equation as E = hν if we want to express this in terms of the frequency of light.
The wave description of light is quite adequate for many applications, but it fails to explain some basic observations. If we shine light (just a form of energy) at a surface of some given material, the energy imparted to the electrons in the material can cause them to jump energy levels or be lost entirely (the atoms are ionized). However, we can only induce electrons to be emitted from the atoms at particular wavelengths of light. Furthermore, increasing the intensity of light of an inappropriate wavelength does not allow electrons to be emitted. These observations suggest that light comes in packages or quanta, some wavelengths having enough energy, and others not, to release the electrons or cause them to change energy levels within an atom. These different energy levels are referred to as quantum levels.
We can think about this in more detail. Electrons can be considered to have a certain energy associated with them. We might say they occupy an energy level. In Figure 3.25a you can see a depiction of this idea for the hydrogen atom. The different energy levels are given values (shown as n ), where the lowest energy ( n = 1) is the “ground state.” You will notice that if we put energy into these electrons, they can be depicted as jumping up energy levels, and we give particular names to these transitions. Electrons that jump from the ground state to any higher state, or drop from a higher state to the ground state (in this case giving off energy) form the “Lyman series.” Those transitions associated with the next level up ( n = 2) are the Balmer series, and so on.

Figure 3.25 The origin of emission and absorption spectra. (a) Energy levels in the hydrogen atom and the transition series associated with the movement of electrons between these levels. Also illustrated are transitions that cause emission lines, absorption lines, and ionization, the latter involving the complete loss of an electron. (b) A depiction of what one sees in a spectrum for different types of electron transitions. (1) A continuous spectrum, (2) an emission spectrum, showing the lines associated with light given off by electrons moving down from one energy state to another, giving lines at particular wavelengths associated with particular gases, and (3) an absorption spectrum. Electrons moving up energy levels, or being ionized, absorb radiation and therefore remove light in a spectrum at given wavelengths corresponding to the energy of the transition. These dark absorption bands are called Fraunhofer lines.
The frequency of light needed to make an electron jump from one energy level to another (resulting in absorption of that energy; Figure 3.25a), say from level n 1to energy level n 2, can therefore be calculated using the following equation:
(3.3) 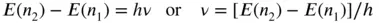
This can be converted to a wavelength using Eq. (3.1). Note that the energy in any given energy level is given as a negative number (Figure 3.25a). Hence the energy of the lower integer energy level (here n 1) is subtracted from the larger integer energy level (here n 2) to get the energy difference between them given in positive units (i.e. in this example, E ( n 2)− E ( n 1) = −3.4 − (−13.6) eV = 10.2 eV required to jump between these two levels.) This can cause confusion, so caution is needed. To avoid confusion, it is best to think about whether energy is being taken in (the electron is moving up energy levels) or being lost as light (the electron is dropping down energy levels and releasing energy in the process).
This equation also tells us what frequency of photon will be emitted when an electron drops an energy level (from n 2to n 1) and emits its energy as a photon. This is also given by ν = [ E ( n 2)− E ( n 1)]/ h . This is conceptually shown in Figure 3.25a as the emission caused when an electron drops an energy level. For example, in the Lyman series for hydrogen, electrons dropping to the ground state give off energy in the ultraviolet (UV) region (and require UV radiation to make them jump from the ground state).
Читать дальше