Halogen Bonding in Solution
Здесь есть возможность читать онлайн «Halogen Bonding in Solution» — ознакомительный отрывок электронной книги совершенно бесплатно, а после прочтения отрывка купить полную версию. В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Жанр: unrecognised, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:Halogen Bonding in Solution
- Автор:
- Жанр:
- Год:неизвестен
- ISBN:нет данных
- Рейтинг книги:5 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 100
- 1
- 2
- 3
- 4
- 5
Halogen Bonding in Solution: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «Halogen Bonding in Solution»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
Halogen Bonding in Solution
Halogen Bonding in Solution
Halogen Bonding in Solution — читать онлайн ознакомительный отрывок
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «Halogen Bonding in Solution», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
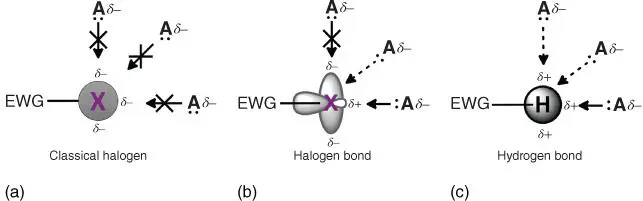
Figure 1.1Schematic of interactions between a halogen atom and a Lewis base acceptor ( A) from a classical view of halogens (a) and from a modern halogen bonding description (b). For comparison the depiction of a hydrogen bond is also included (c). A solid arrow indicates a “stronger” attractive interaction. The dotted arrow is a “less” attractive interaction. The solid arrow with an “X” through it indicates the interaction is repulsive.
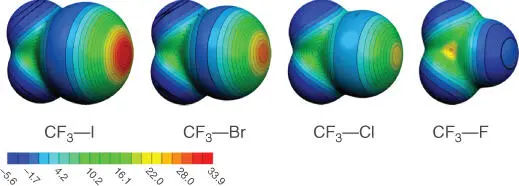
Figure 1.2Molecular electrostatic potential maps drawn at the isodensity surface of 0.001 au for CF 3I, CF 3Br, CF 3Cl, and CF 4. All maps are drawn at the same scale, and values are in kcal/mol.
Source: From Clark et al. [8]. © 2007 Springer Nature.
Disclaimer:It is crucial to note that the σ‐hole description does not account for all the nuances of the halogen bond. Therefore, other conceptual approaches and methodologies (e.g. polarizability, charge transfer) can and should be used to fully describe the halogen bond. These particulars are discussed in the computational section of this chapter. Nevertheless, the σ‐hole is widely used, and ESP maps offer a low barrier to understand the general features and characteristics of the halogen bond.
1.1.2 Parallels to the Hydrogen Bond
The σ‐hole concept elicits obvious parallels between the halogen bond and the hydrogen bond ( Figure 1.1). As such, halogen bond studies, as well as concepts and nomenclature, were undoubtedly inspired by decades of hydrogen bond research. Similarities have prompted frequent comparison between the two, many of which are discussed in the following sections and ensuing chapters. A motivation of these comparative studies has been to discover the limits and unique features of this “new” noncovalent interaction. As such, studies in solution, solid state, and in silico have already demonstrated promising features of the halogen bond compared with its hydrogen analogue.
1.1.3 Notation and Terminology
The vernacular of the halogen bond parallels the hydrogen bond. For example, the hydrogen bond donor references a molecule or group that contains a Lewis acidic hydrogen atom, while the hydrogen bond acceptor is the Lewis basic molecule or group in the interaction. In describing a halogen bond, the donor refers to the molecule with an electrophilic halogen atom (or simply the atom itself, for example, a bromine donor ), and the halogen bond acceptor is the Lewis basic site. Therefore, the common notation for denoting a halogen bond is R–X⋯Y, where Ris a covalently bound species, Xis any halogen with an electrophilic region, Yis the halogen bond acceptor with an electron‐rich region, and ⋯ indicates the attractive noncovalent interaction. Table 1.1highlights common R–Xspecies and several typical Yspecies.
1.1.4 Solid‐state Halogen Bond Contacts
Halogen bond interactions in the solid state are typically quantified by their contact distance and angle with a Lewis base and described using the R–X⋯Ynotation. Additionally, halogen bond contacts are often reported along with a percentage of their combined van der Waals (vdW) radii or more frequently a ratio. The ratio has been given various names such as the halogen bond interaction ratio, normalized interaction distance, normalized contact, or reduction ratio. The ratio is generally defined as 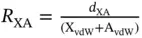 where d XAis the measured distance (Å) from the halogen donor (X) to the acceptor (A), divided by the sum of the vdW radii (Å) of X and A (X vdW+ A vdW). The ratio notation R XAfurther informs the reader, as X is replaced with the atomic symbol of the halogen bond donor, while A denotes the atomic symbol of the halogen bond acceptor atom. For example, R BrOindicates a halogen bond between a bromine donor and an oxygen acceptor. Reporting this ratio enables quick comparison of distances between different halogen bonding sites; yet, it is important to specify the vdW values used as radii can differ based on the sources referenced [9]. Smaller ratio values typically indicate strong halogen bond interactions. When contacts involve anionic Lewis bases, some utilize ionic radii values [10], while others employ vdW radii. Presently, there seems to be no “industry standard,” and simply reporting which values are used is the best practice.
where d XAis the measured distance (Å) from the halogen donor (X) to the acceptor (A), divided by the sum of the vdW radii (Å) of X and A (X vdW+ A vdW). The ratio notation R XAfurther informs the reader, as X is replaced with the atomic symbol of the halogen bond donor, while A denotes the atomic symbol of the halogen bond acceptor atom. For example, R BrOindicates a halogen bond between a bromine donor and an oxygen acceptor. Reporting this ratio enables quick comparison of distances between different halogen bonding sites; yet, it is important to specify the vdW values used as radii can differ based on the sources referenced [9]. Smaller ratio values typically indicate strong halogen bond interactions. When contacts involve anionic Lewis bases, some utilize ionic radii values [10], while others employ vdW radii. Presently, there seems to be no “industry standard,” and simply reporting which values are used is the best practice.
Table 1.1Common halogen bond donors and acceptors.
Source: From Desiraju et al. [2]. © IUPAC.
| Common RX species | Dihalogen molecule (e.g. I 2, Br 2, ICl, ClF)Haloalkane (e.g. CBr 4, CHI 3, C nF 2n+1I)Haloarene or heteroarene (e.g. iodobenzene, halopyridinium, and haloimidazolium cations)1‐Haloalkyne (e.g. diiodoacetylene)Halonium ion (e.g. diphenyliodonium or bromonium derivatives)Haloimide (e.g. N ‐bromo‐ or N ‐iodosuccinimide) |
| Common Y species | Lone pair possessing atom (e.g. N atom of pyridine or an amine, O atom of a carbonyl group) π‐System (e.g. double or triple bonds, arene moiety) Anion (e.g. halides, oxyanions) |
1.1.5 Halogen Bond Features
Characteristics of the halogen bond have been established through experimental and theoretical means. From these studies several features of the halogen bond can be gleaned that should be considered in experimental and functional designs:
The halogen bond is a highly directional interaction. The R–X⋯Y angle tends to be close to 180°. This is due, in part, to the physical characteristics highlighted in Figures 1.1and 1.2.
The halogen bond is highly tunable, with energies up to 200 kJ/mol [11].
Halogens are large atoms resulting in RX bonds, which are longer than R–H counterparts (e.g. vdW radii of 1.46, 1.82, 1.86, 2.04, and 1.20 Å for F, Cl, Br, I, and H, respectively [9]).
Halogen atoms are more hydrophobic than hydrogen atoms and the typical heteroatoms attached to them. Hydrophobicity of halogen atoms is a well‐established phenomenon commonly utilized in drug development where the introduction of a halogen atom into a drug will often result in a drug that is more apt to cross lipid bilayers [12].
Halogen atoms are more polarizable than hydrogens, providing the larger halogen bond donors with a suggested hard–soft acid–base complementarity with soft Lewis bases [13].
Despite the structural differences, both hydrogen and halogen bond donor strength can be tuned similarly by directly altering the donors (substituting heteroatoms and halogens, respectively) and by introducing stronger electron‐withdrawing groups on the R group. Nevertheless, halogen bond tunability is achieved in various ways:
By changing the halogen. A more polarizable halogen will result in a greater σ‐hole (e.g. I > Br > Cl > F). This trend is illustrated in Figure 1.2.
By changing the hybridization of the atom bound to the halogen. For example, with carbon, more s character increases the electron‐withdrawing ability, resulting in a larger σ‐hole on the attached halogen (e.g. C(sp) > C(sp2) > C(sp3)).
By altering the atom, the halogen is bound to (e.g. N–I > C–I).
Читать дальшеИнтервал:
Закладка:
Похожие книги на «Halogen Bonding in Solution»
Представляем Вашему вниманию похожие книги на «Halogen Bonding in Solution» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «Halogen Bonding in Solution» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.












