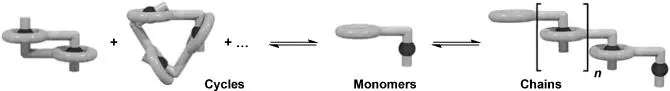
Figure 1.12 Schematic representation of the formation of a poly(pseudorotaxane) via a ring‐chain equilibrium.
Source: Cantrill et al. [95]. © 2001 American Chemical Society.
1.3.3 (Anti)‐cooperative Supramolecular Polymerization
The third and last mechanism for supramolecular polymerization to be discussed herein involves (at least) two distinct stages, resulting in a cooperative or an anti‐cooperative growth of the polymer chains. At first glance, the mechanism of the cooperative supramolecular polymerization is reminiscent to the one for the IDP; however, the polymerization initially occurs via the reversible binding of monomers to the growing chain (as for the IDP, all these steps basically possess the same equilibrium constant K n). At a certain DP, a nucleus is formed and, from this point on, the binding of monomers to the polymer chain features an association constant K e, which is higher than K n( Figure 1.13). In such a nucleation‐elongation polymerization (NEP) model, the supramolecular polymerization proceeds via a linear IDP. In this elongation phase, the actual association constant is now K erather than K n[26, 43, 100, 101].
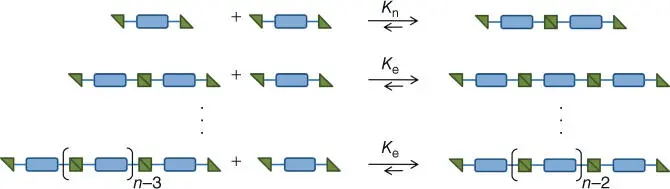
Figure 1.13 Schematic representation of a typical cooperative supramolecular polymerization reaction (nucleation‐elongation mechanism). K nand K erepresent the association constants for the nucleation and the elongation phase, respectively ( K n< K e).
Source: Winter et al. [39]. © 2012 Elsevier B.V.
The complex thermodynamics of the (anti‐)cooperative supramolecular polymerization have already been summarized by de Greef et al.; the reader is referred to this review for a more in‐depth discussion [26]. In the following, a few general aspects concerning the different types of cooperative supramolecular polymerization shall be named. First, one can distinguish between the nucleated and the downhill cooperative supramolecular polymerizations. Ferrone defined a nucleated supramolecular polymerization as a process wherein the initial steps of the chain growth are characterized by an increase of Δ G 0of the oligomers relative to the monomer ( Figure 1.14a) [102]. Beyond the point of nucleation, characterized by a maximum in Δ G 0, polymerization becomes energetically favorable. Now, the nucleus represents the least stable and, thus, the least abundant species in the supramolecular polymerization; as a result, the formation of new polymer chains is retarded (the so‐called bottleneck effect). It is widely accepted that formation of the nucleus occurs via homogeneous nucleation (the analysis of various examples of nucleated supramolecular polymerization has suggested this feature). Noteworthy, however, heterogeneous nucleation is also known, but commonly refers to the nucleation processes on foreign substrates [103]: foreign molecules (e.g. impurities) [104–106], external surfaces (e.g. substrates) [107], dust particles, or secondary nucleation of monomers. The latter eventually give a supramolecular polymer on an already existing one. In particular, the latter has been reported to be dominant in various bio‐supramolecular polymerizations [26].
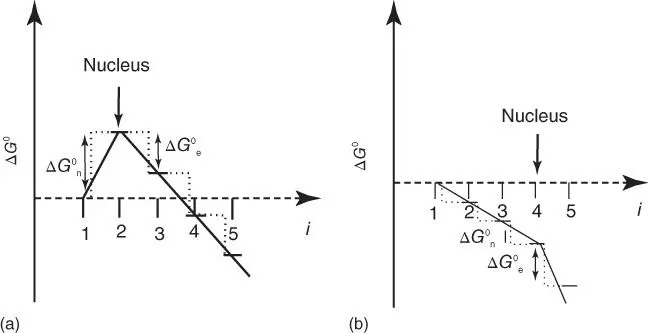
Figure 1.14 Schematic illustration of the energy diagrams of a cooperative nucleated (a) and a cooperative downhill supramolecular polymerization (b). In both plots, the axis of abscissae represents the oligomer's size ( i ), whereas the ordinate measures the Δ G 0in arbitrary units. In diagram (a), the size of the nucleus is 2 (i.e. dimeric nucleus); in diagram (b), a tetrameric nucleus is depicted.
Source: de Greef et al. [26]. © 2009 American Chemical Society.
In summary, three key criteria can be listed according to Frieden to distinguish between an NEP and an IDP [280]:
1 The supramolecular polymerization process is retarded time dependently;
2 This delay of the polymerization can be compensated by adding a preformed nucleus (i.e. seeding); and
3 An equilibrium between the monomer and the supramolecular polymer is established at a certain critical concentration (or temperature).
In contrast to the cooperative nucleated supramolecular polymerization, the cooperative downhill counterpart does not exhibit any increase of Δ G 0in the initial steps. Instead, the initial growth of the polymer is characterized by a lower association constant than the following elongation (i.e. K n> K e; Figure 1.14b). Thus, the monomer is always the species of highest energy in such a cooperative polymerization for which Powers and Powers defined the “nucleus” as the critical chain length at which the absolute (dΔ G 0/d i )‐increment steeply increases [108]. The distinction between the two aforementioned possibilities for cooperative polymerization is associated to the concentration, and, at high total monomer concentrations, a nucleated polymerization process can even be converted into a downhill one [108, 109]. Concentration‐dependent kinetic measurements might, for example, be utilized to distinguish between the two different types of cooperative supramolecular polymerization [108]: in the downhill supramolecular polymerization, the nucleus will be different from the one to be found in a nucleated process (i.e. the nucleus represents a stable or an unstable species, respectively).
For the second type of mechanism, the anti‐cooperative supramolecular polymerization, the initial oligomer formation features an association constant that is much higher than the one for the elongation process. So far, the anti‐cooperative growth in supramolecular polymerizations has attracted less attention, though discrete objects of low dispersity might be obtained (on the contrary, cooperative growth typically gives supramolecular polymers with high Đ values). For example, Mukerjee [110–114] as well as Tanford [115] reported the formation of large aggregates due to the self‐assembly of the surfactants. Due to a high degree of cooperativity in the early stages of the micellar growth, the formation of molecular clusters (i.e. dimers and trimers) was almost fully suppressed. Moreover, (electro)static interactions between the polar head groups of the molecules were identified as the origin of the anti‐cooperative effects, affording micelles of finite size.
What is the covalent counterpart to cooperative supramolecular polymerization? In typical chain‐growth polymerizations (either ionic or radical), the initiation step is analogous to the formation of the nucleus in the NEP; both systems also feature a sequence of propagation steps; however, a conventional termination is generally absent in supramolecular polymerizations. One can still utilize that numerous features of the living ionic or the radical polymerization explain the cooperative supramolecular polymerization: e.g. in both cases, monomers and polymer chains are present halfway through the polymerization [26]. Furthermore, cooperative effects have also been observed in conventional polycondensation reactions: Flory's “principle of equal reactivity” [44] is disobeyed if the electronic properties of the termini are changed in course of the chain growth, in particular when the reactivity of the polymer end groups becomes higher as for the monomer (i.e. the reaction of monomers becomes retarded). The polycondensation of phenyl 4‐( N ‐alkylamino)benzoate in THF under basic conditions, initiated by phenyl 4‐nitrobenzoate, represents one example in this respect [116]: the reactivity of the remote phenyl ester moiety is strongly decreased as soon as the monomer is deprotonated; thus, individual monomers will not react with each other. The monomer anion will then only react with the initiator, thus generating an activated species which, in turn, contains phenyl ester moiety with increased reactivity (relative to the anionic monomer). Consequently, only the activated monomers will react, resulting in a polycondensation which shows a chain growth rather than a step‐growth behavior [117, 118].
Читать дальше















