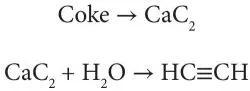Crude oil coke is employed for a number of purposes, but its chief use is in the manufacture of carbon electrodes for aluminum refining, which requires a high-purity carbon – low in ash and sulfur free; the volatile matter must be removed by calcining. In addition to its use as a metallurgical reducing agent, crude oil coke is employed in the manufacture of carbon brushes, silicon carbide abrasives, and structural carbon (e.g., pipes and Rashig rings), as well as calcium carbide manufacture from which acetylene is produced:
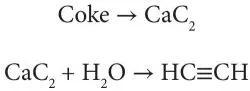
However, with the progressive increase in the amount of coke produced from the heavier (more viscous) refinery feedstocks, there is a renewed interest in the utilization of coke as a feedstock for a gasification process. This is one of the attractive options and is gaining increasing attention to convert the petcoke to value ‐added products. The process offers the refiners a variety of product slates mainly via the production of synthesis gas. The products include steam, hydrogen, electricity, chemicals (such as methanol, CH 3OH, and ammonia, NH 3), and liquid fuels by way of the Fischer-Tropsch synthesis (Murthy et al. , 2013).
The best example of a carbonaceous feedstock for a gasification process is coal, which is often used as a gasification feedstock in a refinery provided suitable coal resources are within economic reach. In fact, as a result of the rapid increase in the use of coal from the 15th century onwards (Nef, 1957; Taylor and Singer, 1957; Speight, 2013) it is not surprising the concept of using coal to produce a flammable gas, especially the use of the water and hot coal (van Heek and Muhlen, 1991), became commonplace (Elton, 1958). However, there are four main types of coal (Table 2.4) and, for maximum process efficiency, the properties of each type of coal must be given due consideration before selection of the requisite gasification process parameters.
Table 2.4The four main types (ranks) of coal *.
| Type/rank |
Properties |
| Lignite Sub-bituminous coal |
Sometimes called brown coal. |
| The youngest of the coal types, and has the lowest energy content. Contains between 25 and 35% w/w carbon. |
| High moisture content coal. |
| Typically burned in power plants for electrical production. |
|
Has a higher energy content than lignite. |
| Contains between 35-45% w/w carbon. |
| Bituminous coal |
Contains anywhere from 45 to 86% w/w carbon. |
| A higher heating value than sub-bituminous coal. |
| Used for electrical production. |
| Plays a large role in the steel and iron industries. |
| Anthracite |
Contains 86 to 97% w/w carbon. |
| Has a slightly lower heating value than bituminous coal. |
*The term low-rank coal refers to the lowest heating value and higher moisture content lignite and sub-bituminous coals.
The production of gas from coal has been a vastly expanding area of coal technology and, as a result, the characteristics of rank, mineral matter, particle size, and reaction conditions are all recognized as having a bearing on the outcome of the process; not only in terms of gas yields but also on gas properties (Massey, 1974; van Heek and Muhlen, 1991). The products from the gasification of coal may be of low, medium, or high heat-content (high-Btu) content as dictated by the process as well as by the ultimate use for the gas (Figure 2.1) (Fryer and Speight, 1976; Mahajan and Walker, 1978; Anderson and Tillman, 1979; Cavagnaro, 1980; Bodle and Huebler; Argonne, 1990; Baker and Rodriguez, 1990; Probstein and Hicks, 1990; Lahaye and Ehrburger, 1991; Matsukata et al. , 1992; Speight, 2013).
Coal is a fossil fuel formed in swamp ecosystems where plant remains were saved from oxidation and biodegradation by being covered with water and mud. Coal is a combustible organic sedimentary rock (composed primarily of carbon, hydrogen and oxygen as well as other minor elements including sulfur) formed from ancient vegetation and consolidated between other rock strata to form coal seams. The harder forms can be regarded as organic metamorphic rocks (e.g., anthracite coal) because of a higher degree of maturation.
Coal remains in adequate supply and at current rates of recovery and consumption, the world global coal reserves have been variously estimated to have a reserves/production ratio of at least 155 years. However, as with all estimates of resource longevity, coal longevity is subject to the assumed rate of consumption remaining at the current rate of consumption and, moreover, to technological developments that dictate the rate at which the coal can be mined. But most importantly, coal is a fossil fuel and an unclean energy source that will only add to global warming. In fact, the next time electricity is advertised as a clean energy source, just consider the means by which the majority of electricity is produced – almost 50% of the electricity generated in the United States derives from coal (EIA, 2007; Speight, 2013).
Coal occurs in different forms or types (Speight, 2013). Variations in the nature of the source material and local or regional variations in the coalification processes cause the vegetal matter to evolve differently. Various classification systems thus exist to define the different types of coal. Chemically, coal is a hydrogen-deficient hydrocarbon with an atomic hydrogen-to-carbon ratio near 0.8, as compared to crude oil hydrocarbon derivatives, which have an atomic hydrogen-to-carbon ratio approximately equal to 2, and methane (CH 4) that has an atomic carbon-to-hydrogen ratio equal to 4. For this reason, any process used to convert coal to alternative fuels must add hydrogen or redistribute the hydrogen in the original coal to generate hydrogen-rich products and coke (Speight, 2013).
The chemical composition of the coal is defined in terms of its proximate and ultimate (elemental) analyses (Speight, 2013). The parameters of proximate analysis are (i) moisture, (ii) volatile matter, (iii) mineral matter, which is determined as combustion ash, and (iv) fixed carbon. Elemental or ultimate analysis encompasses the quantitative determination of carbon, hydrogen, nitrogen, oxygen, and sulfur within the coal. Additionally, specific physical and mechanical properties of coal and particular carbonization properties are also determined.
Carbon monoxide and hydrogen are produced by the gasification of coal in which a mixture of gases is produced. In addition to carbon monoxide and hydrogen, methane and other hydrocarbon derivatives are also produced depending on conditions. Gasification may be accomplished either in situ or in processing plants. In situ gasification is accomplished by controlled, incomplete burning of a coal bed underground while adding air and steam. The gases are withdrawn and may be burned to produce heat, generate electricity or are utilized as synthesis gas in indirect liquefaction as well as for the production of chemicals.
Producing diesel and other fuels from coal can be performed through the conversion of coal to synthesis gas, a combination of carbon monoxide, hydrogen, carbon dioxide, and methane. Synthesis gas is subsequently reacted through Fischer-Tropsch Synthesis processes to produce hydrocarbon derivatives that can be refined into liquid fuels. By increasing the quantity of high-quality fuels from coal (while reducing costs), research into this process could help in mitigating the dependence on ever-increasingly expensive and depleting stocks of crude oil.
Читать дальше