In 1895—the year that Rontgen discovered x-rays—Rutherford borrowed money for his passage to England, packed up his magnetic detector, and set out. Almost immediately on reaching London, he skidded on a banana skin and wrenched his knee. It was several days before he could catch a train to Cambridge and limp into the famous Cavendish Laboratory. His scholarship did not specify which university he should go to. It was up to Rutherford to find a place where he wanted to work and which was willing to accept him. The Cavendish, with its impressive pedigree, seemed a promising possibility.
The laboratory had been founded in the 1870s by William Cavendish, the gifted seventh duke of Devonshire, who, according to an admiring article in Vanity Fair, “ would have been a rare professor of mathematics” had he not been born a nobleman. The first holder of the Cavendish chair of physics had been James Clerk Maxwell, a Scottish laird who in 1 864 had published his theory of electromagnetic fields, showing that electricity and magnetism constituted a single fundamental unity. Taking up his appointment in 1871, he had prophetically warned against the prevailing opinion that “in a few years all the great physical constants will have been approximately estimated, and that the only occupation… left to men of science will be to carry on these measurements to another place of decimals…. we have no right to think thus of the unsearchable riches of creation, or of the untried fertility of those fresh minds into which these riches will continue to be poured.”
The Cavendish and its amiable director, Professor Joseph John Thomson, impressed Rutherford immediately. Known to his students as J. J., Thomson was a Manchester-born mathematician, the son of an impecunious bookseller. In 1884 he had been appointed head of the Cavendish Laboratory at the age of just twenty-eight. His reluctance to pay for elaborate or expensive equipment, perhaps the result of his impoverished childhood, had established the legendary “sealing wax-and-string” tradition of the Cavendish, where everyday materials were ingeniously used to make and patch up experimental equipment, with sealing wax proving particularly useful for vacuum seals. Thomson was, Rutherford noted, badly shaven, with long hair, a small straggling mustache, and a thin, furrowed, clever-looking face. He also had “a most radiating smile” and, at just forty, was “not fossilised at all.”

J. J. Thomson in the Cavendish laboratory
Rutherford decided that he would indeed like to work at the Cavendish. He was fortunate that Cambridge University had just opened its doors for the first time to research students who had graduated elsewhere and was prepared to accept him. With characteristic optimism he hoped he would quickly make enough money from developing his magnetic detector to enable him to marry his fiancée, Mary Newton, the eldest daughter of his erstwhile landlady in Christchurch. Soon he was bustling vigorously around Cambridge, setting up experiments and receiving radio signals from more than half a mile away. As a “colonial,” he was perceived as something of an oddity and was sometimes the object of clumsy jokes, but his robust good humor, undoubted ability, and passion to find things out impressed his colleagues. One wrote with grudging admiration, “We’ve got a rabbit here from the Antipodes and he’s burrowing mighty deep.”
When news of Rontgen’s x-rays reached Cambridge, a greatly excited J. J. Thomson obtained one of the very first x-ray photographs and urged Rutherford to study the phenomenon. He progressively weaned Rutherford away from radio waves, leaving the field of commercial radio development to Guglielmo Marconi, whose work at this time was not as advanced as Rutherford’s. Rutherford began replicating Rontgen’s experiments. The methodology for producing x-rays struck him as very simple, and by the end of 1896 he classed himself as an authority. He was by then working closely with Thomson on explaining how x-rays made gases capable of conducting electricity. He was fascinated by the behavior of the ions—electrically charged atoms—which made this possible. When a colleague cast doubt on their existence, he indignantly replied that ions were “jolly little beggars, you can almost see them.”
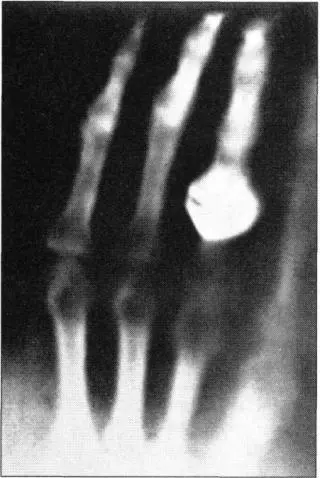
One of the first x-ray images of a human
Reports of Henri Becquerel’s discovery of penetrating rays emitted by uranium salts and of Marie Curie’s experiments with uranium ore roused Rutherford’s curiosity still further. By wrapping uranium in successively increasing layers of thin aluminum foil and observing how the growing thickness of the foil affected the nature and intensity of the escaping radiation, he realized that the uranium was emitting at least two distinct types of radiation. He named them “alpha” and “beta” from the first two letters of the Greek alphabet. Alpha rays could be easily contained, but beta rays, one hundred times more penetrating, could pass through metal barriers. He also believed he detected the presence of a third and highly penetrative radiation—later called “gamma rays” by the Frenchman Paul Villard, who is also sometimes credited with their formal discovery. However, the cause and origin of each of these radiations was, as Rutherford wrote, a mystery which he determined to solve.
At the same time, Rutherford was keen on enjoying Cambridge. With interests far beyond science, he relished the rich texture of university life and, as he wrote to his fiancée, overcame “my usual shyness or rather self-consciousness.” His vigorous intellect attracted people from all fields, including a Hegelian philosopher who invited him to breakfast. The meeting was not, apparently, a success. Rutherford wrote that “he gave me a very poor breakfast, worse luck. His philosophy doesn’t count for much when brought face to face with two kidneys, a thing I abhor.” Rutherford was elected to several exclusive academic clubs, and he had plenty of friends to vacation with. At a seaside resort he was amused when a policeman asked him to swim farther along the beach because the landlady of a boardinghouse opposite objected to the sight of young men in swim suits. He wrote to Mary that “the alarming modesty of the British female is most remarkable—especially the spinster, but I must record to the credit of those who were staying there, that a party of four girls used to regularly do the esplanade at the same hour as we took our dips.”
Meanwhile, Rutherford’s mentor, J. J. Thomson, was about to make the most significant scientific find of the late nineteenth century, a discovery which would profoundly influence Rutherford’s own career. Thomson had been investigating the nature of cathode rays. He was convinced that they were some kind of electrified particles and, to prove his theory, began testing their behavior in electric or magnetic fields. By measuring both the extent to which such fields deflected them and their electrical charge, he discovered that cathode rays consisted of very small negatively charged particles whose mass was about eighteen hundred times less than the lightest known substance—the hydrogen atom. They were, in fact, totally different from an atom. He initially named these tiny carriers of electricity “corpuscles.” Later they would become known as “electrons.”
The corpuscles were, in fact, the first subatomic particles to be found, but their nature was much debated at the time. Their discovery hinted that the atom was not indivisible. Thomson himself admitted that “the assumption of a state of matter more finely subdivided than the atom is a somewhat startling one.” A colleague later told him he thought Thomson had been “pulling their legs.” Thomson’s work suggested an alternative vision—the instability of matter—to that of the indivisible atom. It was revolutionary stuff. Since the seventeenth and eighteenth centuries most leading scientists, including Newton, had believed the atom to be the smallest unit of matter. Some of the ancient Greeks had shared this view—the word atom comes from the Greek atomos, meaning “indivisible.” [6] The ancient Greeks had two theories about the nature of matter. Some, like Aristotle, believed matter was infinite and continuous and so could be infinitely subdivided. Others, like Democritus and Epicurus, thought that matter consisted of minute and indivisible particles.
In the early nineteenth century the English Quaker scientist John Dalton had defined the atomic theory that, by J. J. Thomson’s day, remained the orthodox view. This stated that atoms were the basic and smallest units of matter. Each chemical element consisted of huge quantities of identical atoms. What differentiated the respective elements was only the atoms’ weight and chemical activity. Dalton’s vision of atoms was the Newtonian one of hard, indestructible billiard balls whose arrangement determined the characteristics of chemical compounds.
Читать дальше














