Pierre was soon broaching marriage. Marie hesitated, knowing that it would prevent her cherished scheme of one day returning to her homeland to teach. During a visit to Poland in the summer of 1894, despite her feelings for Pierre, she actively explored the prospect of an appointment at the University of Cracow. However, Pierre knew exactly how to woo her, writing to her that “it would, nevertheless, be a beautiful thing in which I hardly dare believe, to pass through life together hypnotized in our dreams; your dream for your country, our dream for humanity; our dream for science. Of all these dreams, I believe the last, alone, is legitimate.” Such pleas touched Marie, as did his offer to move to Poland, a sacrifice which she told her sister Bronya she had no right to accept. On 26 July 1895 Pierre and Marie were married at a brief civil ceremony with no white dress, wedding ring, or elaborate wedding breakfast. They spent their honeymoon roaming Brittany on bicycles purchased with money given as a wedding present.
By early September the Curies were back in Paris, living in a tiny three-room apartment which Marie, impatient of domestic distractions, furnished with the bare minimum: two chairs, a table, bookshelves, and a bed. Just before their wedding Pierre Curie had been appointed to a new chair of physics, created especially for him, at the Paris School of Physics and Chemistry. Marie was allowed to transfer her work on steels there from the Sorbonne. As a woman working in a laboratory, she was an object of curiosity and some animosity, but this did not deter her. Neither did the birth in September 1897 of the Curies’ first daughter, Irene, whom Marie delightedly called her “little queen” in letters home to Poland. She completed her report on steels within three months of the birth and at once began seeking a suitable subject for her doctoral thesis. She chose a newly discovered subject—Becquerel rays.
• • •
Becquerel rays owed their discovery to a phenomenon that had caught the public imagination. Two years earlier in late 1895, Wilhelm Rontgen, a reclusive German physicist at the University of Würzburg, had been following up work by the Heidelberg physicist Philipp Lenard on how electrical currents pass through gases at low pressures. Rontgen’s prime piece of equipment was a three-foot-long glass tube from which most of the air had been pumped out. Inside the tube were two metal terminals—one positive, called the “anode,” and the other negative, called the “cathode.” Fine wires passing through the glass connected the terminals to an electrical source.
Lenard had observed that when the power was on, the negative plate produced a stream of rays which caused the tube walls to glow with a soft green light. Rontgen was prepared for this. What startled him was that, despite the black card with which he had mantled his tube to exclude exterior influences on his observations, a nearby paper screen painted with fluorescent substances (barium platinocyanide) was also glowing brightly. In fact, each time electricity pulsed through the blacked-out tube, the paper screen luminesced. Rontgen moved the screen two yards away from the tube, but still it glowed.
Lenard’s experiments had demonstrated that cathode rays were stopped by quite thin barriers, so Rontgen realized that some sort of penetrating rays—hitherto unknown and which he therefore named “x-rays”—were escaping through the glass walls of his tube. He further deduced that these x-rays were caused by the impact of the cathode rays on the tube’s glass walls. He discovered that although his x-rays could penetrate thick books or decks of cards, they could not pass through denser materials like metal so easily. When he placed his hand between the tube and the fluorescent screen, Rontgen was staggered to see the shadows of his own bones. The rays had penetrated the soft tissue, but the denser bones were sharply delineated on the screen.
Rontgen tested the rays’ effects using photographic plates, capturing in the world’s first x-ray pictures images of everything from a compass needle in a metal case to his bones. Rontgen realized the implications: His rays could be used to identify fractures in bones and find bullets embedded in tissue. In January 1896 he announced his discovery publicly in Berlin, and before the month was out radiographs were being produced around the world. In 1901 he would become the first recipient of the Nobel Prize for Physics, introduced that year after Alfred Nobel left the bulk of his estate in trust for the annual award of five prizes for services to physics, chemistry, medicine, literature, and peace. In the years ahead, the physics and chemistry awards would be dominated by those exploring the new atomic science.

Punch cartoon of Rontgen’s x-rays
As news of the miraculous rays spread and they were successfully put to work in medical diagnosis, Rontgen became a reluctant celebrity, forced to dodge newspaper reporters. Some people, though, were disturbed by his discovery. Women seriously contemplated buying “x-ray proof underwear” to repel lascivious Peeping Toms. One rhyme warned:
I hear they’ll gaze
Through cloak and gown—and even stays
Those naughty, naughty Rontgen rays
Punch magazine quipped:
We do not want, like Dr. Swft,
To take our flesh off and to pose in
Our bones, or show each little rift
And joint for you to poke your nose in.
We only crave to contemplate
Each other’s usual full-dress photo;
Your worse than “altogether” state
Of portraiture we bar in toto.
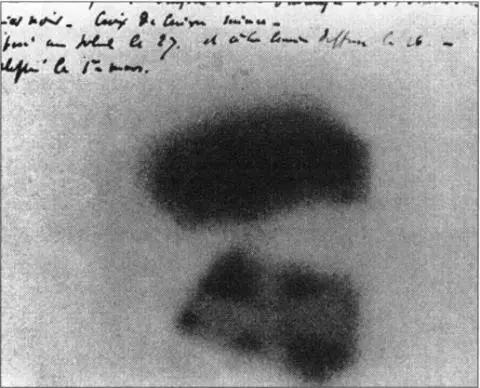
Becquerel’s smudges showing the first proof of radioactivity
Meanwhile, puzzled scientists struggled to explain the source of the mysterious x-rays. In Paris, the physicist and professor Henri Becquerel decided to investigate whether phosphorescent and fluorescent substances produced these invisible rays. [5] Fluorescent substances absorb light of one color or wavelength and in its place radiate light of another color. When the source of the light is turned off, that radiation ceases. With phosphorescent materials, the radiation continues after the light source has been removed.
Becquerel carefully placed successive glowing materials on photographic plates that he had previously wrapped in thick black paper to see whether rays would penetrate the paper and darken the plates. Nothing happened until he selected the powdery white salts of the rare metal uranium, luminous in sunlight. At last, there was a result. When the plates were developed, Becquerel noted faint smudges—evidence of penetrating radiation. He conducted further tests, sometimes adding a coin or metal sheet and observing the faint traces of their outline.
One day he placed uranium salts, together with a copper cross, on a photographic plate, but the Paris weather became overcast. Sharing the common belief that substances needed natural sunlight to luminesce, he thrust the plate into a drawer to await a brighter day. Some days later, on 1 March 1896, sheer chance or what another scientist William Crookes—who was present and saw what happened—admiringly called “the unconscious pre-vision of genius” caused Becquerel to develop the plate. He found that despite being in darkness the uranium salts had emitted radiation. The image of the copper cross was “shining out white against the black background.”
Читать дальше














