Marlene Parrish - What Einstein Told His Cook 2
Здесь есть возможность читать онлайн «Marlene Parrish - What Einstein Told His Cook 2» весь текст электронной книги совершенно бесплатно (целиком полную версию без сокращений). В некоторых случаях можно слушать аудио, скачать через торрент в формате fb2 и присутствует краткое содержание. Год выпуска: 0101, Издательство: W. W. Norton & Company, Жанр: Кулинария, на английском языке. Описание произведения, (предисловие) а так же отзывы посетителей доступны на портале библиотеки ЛибКат.
- Название:What Einstein Told His Cook 2
- Автор:
- Издательство:W. W. Norton & Company
- Жанр:
- Год:0101
- ISBN:нет данных
- Рейтинг книги:4 / 5. Голосов: 1
-
Избранное:Добавить в избранное
- Отзывы:
-
Ваша оценка:
- 80
- 1
- 2
- 3
- 4
- 5
What Einstein Told His Cook 2: краткое содержание, описание и аннотация
Предлагаем к чтению аннотацию, описание, краткое содержание или предисловие (зависит от того, что написал сам автор книги «What Einstein Told His Cook 2»). Если вы не нашли необходимую информацию о книге — напишите в комментариях, мы постараемся отыскать её.
What Einstein Told His Cook 2 — читать онлайн бесплатно полную книгу (весь текст) целиком
Ниже представлен текст книги, разбитый по страницам. Система сохранения места последней прочитанной страницы, позволяет с удобством читать онлайн бесплатно книгу «What Einstein Told His Cook 2», без необходимости каждый раз заново искать на чём Вы остановились. Поставьте закладку, и сможете в любой момент перейти на страницу, на которой закончили чтение.
Интервал:
Закладка:
No, just kidding about O.J.
Trans fatty acids (I’ll call them “trans FA’s” from here on) don’t occur naturally, except for small amounts in a few plants such as pomegranates, cabbage, and peas and making up about 3 to 5 percent of the fatty acids in the meat and milk of ruminants: cows, sheep, and goats. They are created in much larger amounts during the artificial hydrogenation of vegetable oils to make them more solid, most often to convert liquid soybean oil into manageably spreadable margarines. In fact, trans FA’s are in every food that says “partially hydrogenated vegetable [or the name of a specific vegetable] oil” in the list of ingredients on the label. And you can assume that virtually everything on the snack-food shelves of your local convenience store is loaded with trans FA’s.
Understanding trans FA’s requires digesting a bit of chemistry. I have set this information aside in “Kinky molecules” on chapter 4. You may think of it as what the textbooks call Further Reading, which of course nobody reads. Read it or not; it’s your call.
A no-trans tip
The softer a margarine is, the less it has been hydrogenated and the less trans fatty acid it will contain. But I don’t like soft, almost liquid margarines; I like them to have some firmness for spreading on my toast. So I buy a soft, no-trans-fat margarine (according to the label) and keep it in the freezer, where it firms up to a perfect spreading consistency.
With or without the FDA’s required, mandatory, compulsory, and obligatory labeling, how can you tell where all the trans FA’s are hiding? You’re not going to like this, but partially hydrogenated fats carrying their burden of trans FA’s lurk in virtually everything you love to eat: margarine, commercial cakes and cookies, doughnuts, potato chips, crackers, popcorn, nondairy creamers, whipped toppings, gravy mixes, cake mixes, frozen French fries and pizzas, fish sticks, and virtually all commercially fried foods.
Restaurants that brag about using only “pure vegetable oil” don’t tell you that it may contain as much as 40 percent trans FA’s. Peek into the kitchen and you may see that before it was melted it was delivered as a semisolid, like Crisco. That’s the tip-off that it has been hydrogenated—had hydrogen gas forced into it at a high temperature and pressure. (Unless that bucket of white fat is lard, which is another whole story.) To make matters worse, trans FA’s are formed in small amounts at the high temperatures of frying, so you may even be producing them yourself at home.
There is, however, a ray of hope. The amount of trans FA’s formed in the hydrogenation of oils depends on the temperature, the hydrogen gas pressure, the length of exposure, and many other factors. Now that the pressure is on from the feds, you can bet your Twinkie that packaged-food manufacturers have been scrambling to find ways of attaining the desired physical characteristics in their fats with the minimum production of trans FA’s. They want to earn the right to put the coveted phrase “Contains no trans fatty acids” or “Contains no trans fats” on their labels.
But note that “no trans fatty acids” on a label doesn’t mean NO trans fatty acids. According to FDA labeling regulations, it means less than 0.5 gram per serving. Insisting that there not be a single molecule of trans fatty acid in a food would be both unrealistic and unenforceable. There has to be some upper-limit definition of “none.”
Oh, and about fractionated oils: Not to worry. Fractionation has nothing to do with trans FA’s. All it does is remove some of the more saturated, higher-melting fats to keep the product from thickening or freezing when stored in a cool place.
Sidebar Science: Kinky molecules
A MOLECULEof any fat (a triglyceride ) contains three fatty acid molecules attached to a glycerol (glycerin) base. The three fatty acids can be any combination of saturated, monounsaturated, or polyunsaturated. The health consequences of any given fat are purely those of the fatty acids (I’ll call them FA’s from here on) it contains.
The FA parts consist almost entirely of long chains of carbon atoms with hydrogen atoms sticking out like hairs on a caterpillar. In a molecule of a saturated FA, every carbon atom in the chain carries two hydrogen atoms, so the chains look like this: –CH 2–CH 2–CH 2–CH 2–, etc. (C represents a carbon atom, H represents a hydrogen atom, and—represents a chemical bond between carbon atoms.)
But in an unsaturated FA, there are occasional locations where two adjacent carbon atoms have only one hydrogen atom apiece, and the chain looks like this: –CH 2–CH=CH–CH 2–, etc. The two middle carbon atoms have squandered twice as much of their available bonding power just between themselves, with none left over for grabbing another two hydrogen atoms. That kind of connection between carbon atoms is called a double bond, indicated by =. If there is one such location in a FA molecule, it is said to be monounsaturated; two or more make it a polyunsaturated FA.
Wherever a double bond occurs in an unsaturated FA, it makes a kink or bend in the otherwise straight chain. Kinky molecules can’t pack together as closely as straight molecules can, so the molecules are looser and an unsaturated fat tends to be a runny liquid rather than a firm, compact solid.
But even more significant than the physical properties of the fat is the fact that in most biological processes the exact shapes of molecules can be enormously important. As we metabolize them, it is primarily their different shapes that make kinky, unsaturated FA molecules more healthful than straight, saturated ones.
Food manufacturers want to convert unsaturated liquid fats into semisolid, consumer-friendly fats. So by applying high hydrogen gas pressure and heat—up to 150 psi (10 atmospheres) and 430°F (220°C)—they force two more hydrogen atoms into the double bonds. That is, they hydrogenate the unsaturated FA’s to make them more saturated. But if they were to saturate every double bond in a polyunsaturated FA, it would become so hard that it would be as inedible as candle wax.
That’s why liquid vegetable oils are only partially hydrogenated—that is, only a fraction of their double bonds are filled in with hydrogen atoms. Moreover, the hydrogenation process is inherently inefficient, so complete hydrogenation would be difficult to achieve anyway.
But here’s where the trans FA’s come in. During the course of hydrogenation, some of the double bonds evade the addition of two more hydrogen atoms by skipping off to another part of the chain. (The double bond migrates .) In the process, their original two hydrogen atoms, circled in the illustrations, which may have been on the same side of the double bond—in a cis configuration —are likely to flip to opposite sides—into a trans configuration. This hydrogen-atom-flipping can take place even without double-bond migration, because the trans form is inherently more stable than the cis form. ( Cis , pronounced “sis,” and trans are from the Latin, meaning “on this side” and “across,” respectively.)
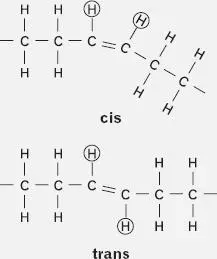
So what if the hydrogen atoms do switch from the cis to the trans positions? Well, the two different resulting molecules (the two so-called isomers ) have the same number of atoms of each kind, but they have different shapes. The cis molecules retain the original kinky shape of a normal unsaturated FA. But the newly formed trans molecules are straighter, more resembling a saturated FA.
Читать дальшеИнтервал:
Закладка:
Похожие книги на «What Einstein Told His Cook 2»
Представляем Вашему вниманию похожие книги на «What Einstein Told His Cook 2» списком для выбора. Мы отобрали схожую по названию и смыслу литературу в надежде предоставить читателям больше вариантов отыскать новые, интересные, ещё непрочитанные произведения.
Обсуждение, отзывы о книге «What Einstein Told His Cook 2» и просто собственные мнения читателей. Оставьте ваши комментарии, напишите, что Вы думаете о произведении, его смысле или главных героях. Укажите что конкретно понравилось, а что нет, и почему Вы так считаете.